Molecules 2013, 18
436
1
2
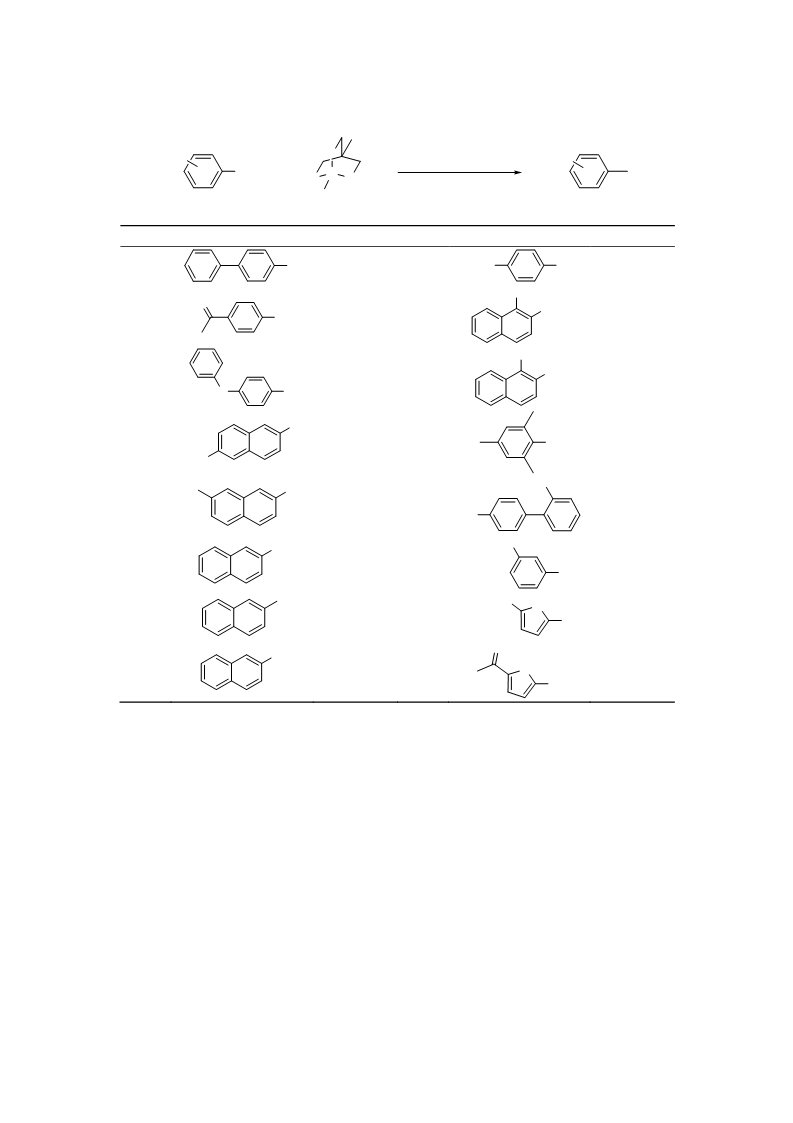
-Methylnaphthalene (entries 6–8): H-NMR (CDCl ) δ = 7.82–7.72 (m, 3H), 7.67 (s, 1H), 7.51–7.40
3
(
m, 2H), 7.33 (d, J = 8.15 Hz, 1H), 2.57 (s, 3H).
1
1
-Methyl-4-nitrobenzene (entry 9): H-NMR (CDCl
3
) δ = 8.10 (d, J = 8.61 Hz, 2H), 7.31 (d, J = 8.61 Hz,
2H), 2.45 (s, 3H).
1
2
-Methoxy-1-methylnaphthalene (entry 10): H-NMR (CDCl ) δ = 8.03 (d, J = 7.89 Hz, 1H), 7.86 (d,
3
J = 8.25 Hz, 1H), 7.78 (d, J = 8.97 Hz, 1H), 7.56 (ddd, J = 1.43, 6.82, 8.32 Hz, 1H), 7.43 (ddd, J = 1.08,
6
.67, 7.44 Hz, 1H), 7.32 (d, J = 8.97 Hz, 1H), 3.99 (s, 3H), 2.65 (s, 3H).
1
1
,2-Dimethylnaphthalene (entry 11): H-NMR (CDCl
3
) δ = 8.08 (d, J = 8.61 Hz, 1H), 7.85 (d, J = 7.89
Hz, 1H), 7.67 (d, J = 8.25 Hz, 1H), 7.54 (ddd, J = 1.43, 6.82, 8.34 Hz, 1H), 7.46 (t, J = 6.82 Hz, 1H),
7
1
2
.35 (d, J = 8.25 Hz, 1H), 2.65 (s, 3H), 2.54 (s, 3H).
1
,2,3,5-Tetramethylbenzene (entry 12): H-NMR (CDCl
3
) δ = 6.88 (s, 2H), 2.29 (s, 9H), 2.18 (s, 3H).
1
'-Methylbiphenyl-4-carbonitrile (entry 13): H-NMR (CDCl ) δ = 7.72–7.68 (m, 3H), 7.44–7.41
3
(
m, 2H), 7.32–7.25 (m, 2H), 7.19 (d, J = 7.17 Hz, 1H), 2.26 (s, 3H).
1
Methyl 3-methylbenzoate (entry 14): H-NMR (CDCl
3
) δ = 7.85–7.82 (m, 2H), 7.36–7.29 (m, 2H),
3.89 (s, 3H), 2.38 (s, 3H).
1
Methyl 5-methylfuran-2-carboxylate (entry 15): H-NMR (CDCl
d, J = 3.23 Hz, 1H), 2.34 (s, 3H).
3
) δ = 7.05 (d, J = 3.59 Hz, 1H), 6.08
(
1
2
3
-Acetyl-5-methylthiophene (entry 16): H-NMR (CDCl ) δ = 7.48 (d, J = 3.59 Hz, 1H), 6.76 (dd, J = 1.08,
3
.77 Hz, 1H), 2.50 (s, 3H), 2.41 (s, 3H).
4
. Conclusions
In summary, we have demonstrated the efficiency of lithium methyltriolborate for methylation of
aryl halides. This borate showed several advantages over boronic acid, including high nucleophilicity
of methyl groups for smooth transmetalation to a palladium catalyst. Since the use of a base is avoided,
a variety of functional groups may be accommodated in this reaction system.
Acknowledgments
This work was supported in part by Strategic Molecular and Materials Chemistry through Innovative
Coupling Reactions from the Ministry of Education, Culture, Sports, Science, and Technology, Japan.
References
1
.
Miyaura, N.; Suzuki, A. Palladium-Catalyzed Cross-Coupling Reactions of Organoboron
Compounds. Chem. Rev. 1995, 95, 2457–2483.

 Yamamoto, Yasunori
Yamamoto, Yasunori
 Ikizakura, Kazuya
Ikizakura, Kazuya
 Ito, Hajime
Ito, Hajime
 Miyaura, Norio
Miyaura, Norio