Chem. Pap.
metal chelating ability and has unique advantages in the
design of iron ions recognition probes (Ali et al. 2011;
Brombosz et al. 2007). However, in the previous reports,
most fluorophores detecting Fe2? suffer from the interfer-
ence of other metal ions, especially Fe3?. Recently, a
CdSO4Á8H2O, BaCl2Á2H2O, Hg(ClO4)2Á3H2O, and
Pb(NO3)2 were prepared by dissolving them in double
distilled water. All samples were prepared at room tem-
perature. The emission spectra were recorded when excited
at 430 nm. All the measurements were carried out at room
temperature.
graphene oxide-terpyridine conjugate as
a
nano-
chemosensor for Fe2? in aqueous media was reported
(Eftekhari-Sis and Mirdoraghi 2016), nevertheless, specific
terpyridine functionalized Fe2? probe was rare according
to our investigation. Our group managed developing two
kinds of terpyridine functionalized a-cyanostilbene
derivatives which detected Fe2? in aqueous environment
(Liang et al. 2007; Zhang et al. 2014). Herein, we imagined
to introduce carbazole unit, a famous fluorophores with
high luminescence efficiency, hole-transporting moiety
into the system to design a selective Fe2? probe with AIEE
properties.
The synthetic route of compound L was shown in Fig. 1.
Compound 1 was prepared according to the literature
methods (Jin et al. 2011; Constable and Ward. 1990; Potts
and Konwar. 1991).
9-(4-Bromobutyl)-9H-carbazole (2): a mixture of car-
bazole (3.3 g, 20 mmol), 1,4-dibromobutane (20 mL),
toluene (15 mL), and tetrabutylammonium bromide 1.0 g
in 15 mL 50% aqueous sodium hydroxide solution were
stirred at 45 °C for 3 h, and then cooled to RT. The mixture
was stirred overnight. After extraction with CH2Cl2, the
organic phase was washed by water and saturated salt
water for three times, respectively, and dried with anhy-
drous sodium sulfate. The solvent was removed under
reduced pressure. The crude product was recrystallized
from ethanol to obtain a white powder (3.83 g, 66.3%
yield).
In this work, a novel carbazole derivative with func-
tionalized terpyridine (L) was synthesized. The photo-
physical properties of L were studied by UV–vis
absorption spectroscopy and fluorescence spectroscopy.
This sensitive probe exhibited excellent AIEE property and
could be used to detect single Fe2? in aqueous solution.
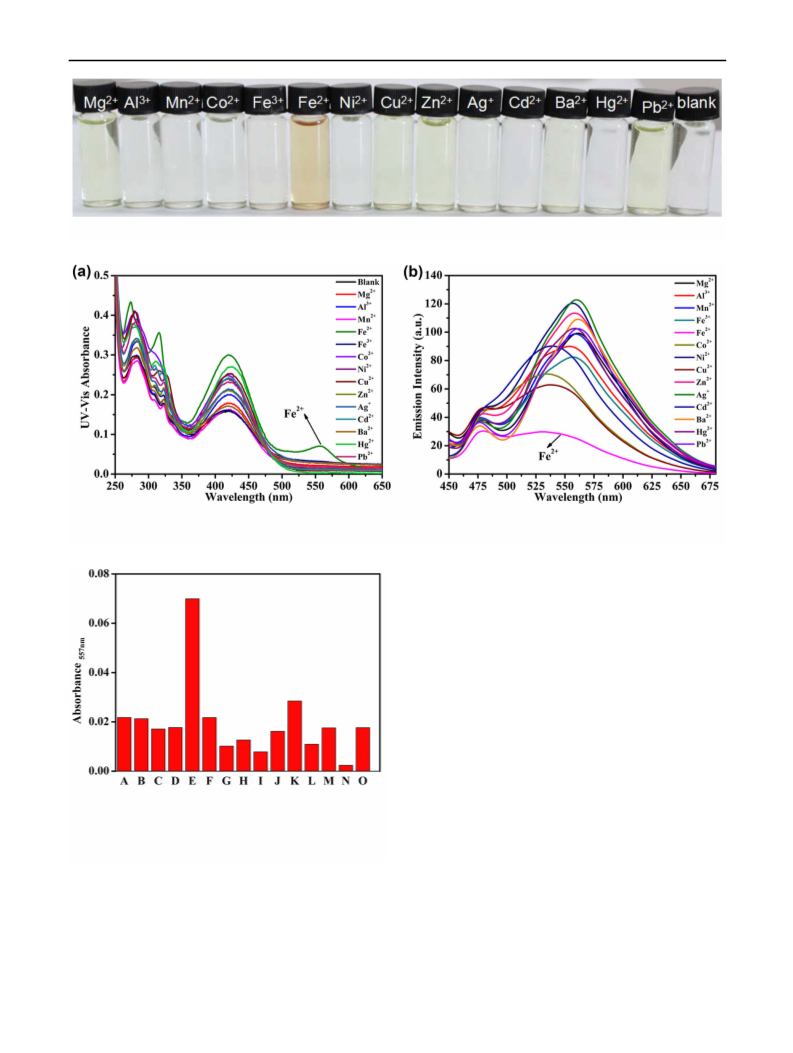
Other metal ions including Fe3? caused little interference.
Fortunately, L could act as a dual mode Fe2? sensor via
UV–vis absorption spectroscopy and ‘‘naked eyes’’
recognition.
9-(3-Bromopropyl)-9H-carbazole-3-carbaldehyde (3):
5.4 mL DMF (70 mmol), 6.4 mL (70 mmol) POCl3 were
cooled to 0 °C by an external ice bath. Later, compound 2
(7.0 g, 23.2 mmol) dissolved in 1,2-dichloroethane was
added to the mixture. The mixture was stirred at 80 °C
overnight, then, it was added to ice water. After neutral-
ization to weak basicity by NaOH, the solution was
extracted with CH2Cl2. The organic layer was dried over
with anhydrous sodium sulfate and filtered. The solvent
was removed under reduced pressure. The residue was
purified by silica gel column chromatography (eluent:
CH2Cl2) to afford a white solid (4.29 g, 56.1% yield). m.
Experimental
All chemicals were commercially available and all solvents
were purified by conventional methods before used. NMR
spectra were recorded on a Bruker Avance 400 MHz
spectrometer using CDCl3 or DMF-d7 as solvent. Chemical
shifts were reported in parts per million (ppm) down field
from TMS with the solvent resonance as the internal
standard. Coupling constants (J) were reported in Hz and
referred to apparent peak multiplications. Mass spectra
were recorded on an Agilent 6410 LC–MS/MS system
(USA) equipped with an electrospray ion source (ESI).
UV–vis absorption spectra were recorded on a TU-1901 of
Beijing Purkinje General Instrument Co., Ltd, spectrometer
(slit: 5 nm, 3 nm) using samples in solutions. Fluorescence
spectra were recorded with a RF-5301 PC fluorescence
spectrometer. The stock solution (1 9 10-3 M) was pre-
pared by dissolving probe L in DMF. For the UV–vis and
fluorescence analysis, 50 lL of the stock solution of L in
DMF was diluted to 5 mL DMF-H2O mixture or EtOH-
H2O mixture (volume fraction of water accounts for 30%),
respectively. Solutions of MgSO4, Al2(SO4)3Á18H2O,
MnSO4ÁH2O, FeSO4Á7H2O, FeCl3Á6H2O, CoCl2Á6H2O,
NiCl2Á6H2O, CuSO4Á5H2O, ZnSO4Á7H2O, AgNO3,
1
p. 83–85 °C; H NMR (CDCl3, 400 MHz) d 1.828–1.898
(2H, m, CH2), 2.067–2.140 (2H, m, CH2), 3.532–3.563
(2H, t, J = 6.0 Hz, CH2–O), 4.391–4.427 (2H, t,
J = 7.2 Hz, CH2–N), 7.331–7.367 (1H, t, J = 7.2 Hz,
phenyl–H), 7.459–7.498 (2H, t, J = 8.0 Hz, phenyl–H),
7.537–7.575 (1H, t, J = 7.6 Hz, phenyl–H), 8.019–8.040
(1H, d, J = 8.4 Hz, phenyl–H), 8.164–8.184 (1H, d,
J = 8.0 Hz, phenyl–H), 8.627 (1H, s, phenyl–H), 10.112
(1H, s, aldehyde–H); 13C NMR (CDCl3, 100 MHz):
26.356, 29.987, 42.676, 44.341, 108.826, 109.263,
120.506, 120.842, 123.066, 123.187, 123.949, 126.862,
127.312, 128.727, 141.044, 143.931, 191.688.
N-(2,20:6,200,-4- terpyridyl oxybutyl)-3-aldehyde car-
bazole (4): Compound 1 (145.0 mg, 484.5 lmol), com-
pound 3 (160.0 mg, 484.5 lmol), and K2CO3 (80.4 mg,
581.7 lmol) were added in 4 mL acetonitrile and refluxed
overnight. The hot mixture was filtrated. The residue was
washed with acetonitrile and water. After using flash
chromatography (eluent: CH2Cl2) to remove compound 3,
123

 He, Xuan
He, Xuan
 Zhang, Gao-Bin
Zhang, Gao-Bin
 Chi, Zi-Xin
Chi, Zi-Xin
 Dai, Peng-Fei
Dai, Peng-Fei
 Huang, Jian-Yan
Huang, Jian-Yan
 Yang, Jia-Xiang
Yang, Jia-Xiang