H. Xin et al. / Catalysis Communications 93 (2017) 1–4
3
dihydroxybiphenyl under inert atmosphere and combines with oxygen
to form p-benzoquinone under O atmosphere in the absence of I . Ob-
viously, oxygen is a radical scavenger in the absence of I . In the pres-
ence of I and O , I preferentially reacts with phenol and oxygen
2
2
2
2
2
2
plays the role of an oxidant. Although nitrate anions are not active cen-
ters for phenol, they significantly accelerate the reactions. Thus, we con-
sider nitrate anion is likely a co-catalyst, which possibly activate I
2
to
promote the reactions.
The optimized results are listed in Table 2S (see Supporting informa-
tion). When I was replaced with HI, reactions also proceeded smoothly,
2
and a yield of 88% was achieved for 1b in 12 h. As dioxygen was replaced
with air, a high yield of 91% was obtained at 80 °C for 10 h. Studies indi-
cate that common organic solvents, such as methanol, are also effective,
but water is undoubtedly the best alternative. Based on the optimized
conditions, complete conversion was achieved, and a high yield of 93%
for 1b was obtained with 5 mol% of catalyst at 80 °C for 3 h. Because io-
dine is easy to sublimate, part of iodine is always out of the reaction sys-
tem and aggregates as iodine bulk on the top of glassware. The
aggregated iodine bulk was collected, and it is around 1.20–1.25 mmol
according to titration test. Thus, the iodine involved in the reactions is
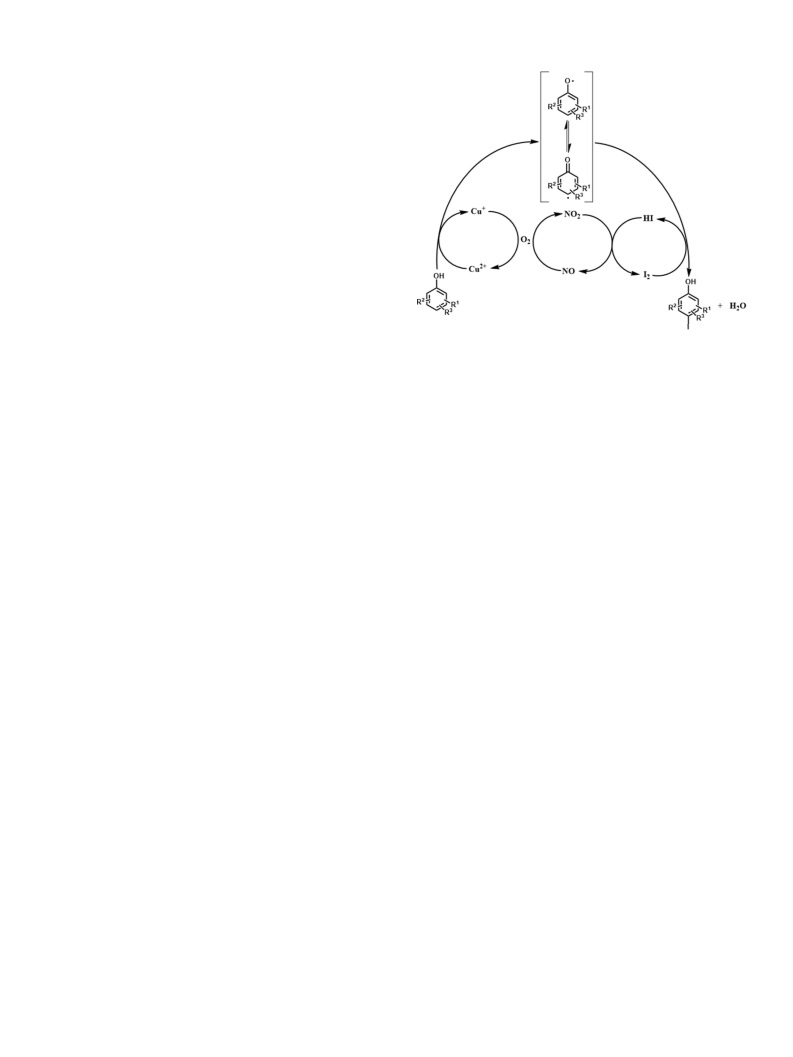
Fig. 1. A possible mechanism of the aerobic oxyiodination of aromatic phenols catalysed by
copper(II) nitrate.
0
.95–1.00 mmol. Since the substrate of 1a is 2.0 mmol, the utilization
of iodine atoms is ≥93% and iodine involved in the reactions is almost
quantitatively incorporated into the product. Therefore, the current ap-
proach demonstrates a high atom economy. When we enlarged the re-
action scale to 50 times in the absence of any VOCs, a conversion of 1a
was 95% with a yield of 1b at 90%. The reaction proceeded well without
VOC (see Supporting information).
are indirect evidences, we think phenoxy radical involves in our system.
Besides those results, the following evidences also support this
hypothesis: 1) when 2,3,6-tri-methylphenol was treated with 1.0 eq.
2
of copper(II) nitrate in the absence of I under Ar atmosphere, a
In further experiments, various substituted phenols were examined
under similar conditions (Table 2). In fact, phenols with electron donat-
ing groups at meta-position can be easily iodinated, and para-
iodophenols were produced in good yield in 3 h (Table 2, run 2–4). A
yield of 82% was achieved for 2b from m-cresol (2a), and a 90% yield
for 3b was obtained from 3,5-dimethylphenol (3a). As for 2,3,5-tri-
methylphenol (4a), the yield of 4b reached as high as 91%. When phe-
nols with electron donating groups grafted at ortho-position were
used, no reaction was observed even the reaction time was prolonged
to 6 h. In order to increase the solubility of these phenols in water, the
phase-transfer reagent, such as tetra-butyl ammonium bromide, was
added into the system. Iodination proceeded smoothly and the reaction
was completed after 6 h (Table 2, run 5–9). A yield for 5b was 83% from
o-cresol (5a), and a yield of 78% for 6b was obtained from 2-tert-
butylphenol (6a). The yield of 7b was 88% from 2,6-dimethylphenol
yield of 60% was detected for 2,2″,3,3″,5,5″-hexamethyl-4,4″-
dihydroxybiphenyl; 2) if a free radical scavenger, 2,6-di-tert-butyl-4-
methyl phenol, was added into the above reactions, almost no products
were found in the reactions; 3) as we took a little solution (about 1 ml)
from the reaction mixtures as the reaction ran for 1.5 h, and performed
−
the precipitation reaction using silver nitrate as a precipitator for I ,
precipitation of silver iodide was observed. Thus, although the details
of reaction mechanism are not clear so far, a possible mechanism is
shown in Fig. 1: Cu2 oxidizes phenols to produce phenoxy radicals,
+
which react with I
NO to I with the formation of NO; and the reduced Cu and NO are
re-oxidized by dioxygen back to Cu and NO
tion is recycled as above described until I is exhausted.
2
to form p-iodophenol and HI; HI is oxidized by
+
2
2
2
+
2
, respectively. The reac-
2
3. Conclusion
(7a). As for 2,6-di-isopropylphenol (8a) and 2,6-di-tert-butylphenol
9a), the yield for 8b and 9b reached 84% and 89%, respectively. Com-
(
A simple, green, and efficient method has been developed for aerobic
pared with unsubstituted phenol (1a) or phenols with electron donat-
ing groups at meta-position, the reaction speed of phenols with
electron donating groups at ortho-position is was slower under the
same conditions. Phenols with electron withdrawing groups can also
be successfully iodinated in our system (Table 2, run 10–12). Iodination
of 2-chlorophenol (10a) gave 10b in 89% yield, and the yield of 11b from
oxyiodination of aromatic phenols using dioxygen and iodine under or-
ganic solvent-free conditions. Copper(II) nitrate showed very high cata-
lytic activity and selectivity for the direct iodination of various phenols
to para-iodophenols with dioxygen in water. A wide range of phenol de-
rivatives were tested, containing either electron-donating or electron-
withdrawing groups, which suggested that this method is suitable to
synthesize various para-iodophenols.
2
-bromophenol (11a) was 86% for 10 h. While 2,4-dichlorophenol
12a) was iodinated as 12b in a high yield of 85% in 12 h, no iodination
was observed for 4-chlorophenol (13a) and 4-bromophenol (14a)
Table 2, run 13–14). Compared with unsubstituted phenol (1a) or phe-
(
Appendix A. Supplementary data
(
nols with electron donating groups, the iodination of phenols with elec-
tron withdrawing groups proceeded at slower speed under the same
conditions. In above reactions, only trace of ortho- or meta-isomer was
observed, and no multi-iodo substituted products were found. The pref-
erential formation of para-iodo substituted phenols by our method is re-
markably selective.
It is known that the oxidative transformations of phenols catalysed
by copper compounds usually involve the oxidation of phenolate by
Cu(II) to phenoxy radical [31]. However, phenoxy radicals are extreme-
ly active and the reaction proceeds too fast to observe the intermediate,
our attempts to obtain the electron paramagnetic resonance of phenoxy
radicals were failed. Although the results shown in control experiments
References
[
[
[
[
[

 Xin, Hongchuan
Xin, Hongchuan
 Hu, Liangning
Hu, Liangning
 Yu, Jianqiang
Yu, Jianqiang
 Sun, Wenshou
Sun, Wenshou
 An, Zengjian
An, Zengjian