8
402 J. Phys. Chem. B, Vol. 108, No. 24, 2004
Goa et al.
carbonate severely lowered the turnover number in the oxidation
of hydrocarbons,2 and TS-1 synthesized in the presence of large
amount of alkali and alkaline earth ions showed low oxidation
activity.22 While acid sites were blocked with these metal cations
and acid-catalyzed solvolysis of the epoxide was suppressed to
show high epoxide selectivity, these cations simultaneously
poisoned the active sites for oxidation. As far as we know,
however, the mechanism of the poisoning of titanosilicates by
alkali cations is not clarified yet. Tatsumi et al. have suggested
that SisOsM (M: metal cation) is less covalent and more ionic
than SisOsH; the polar environment around the Ti sites makes
the interaction between the catalyst and the reactant molecules
having CdC double bonds weaker. We have communicated
recently that ion exchange with quaternary ammonium cations
dramatically enhances the epoxide selectivity of [Ti,Al]-Beta
even in the protic solvent alcohols without retarding the specific
activity of Ti species.23 In this study, we have compared this
postsynthetic method with the direct calcinination of the as-
synthesized [Ti,Al]-Beta to reveal the advantage of the postsyn-
thesis on [Ti,Al]-Beta over the direct method. A mechanistic
consideration has been given to how these methods controlled
the acidity of [Ti,Al]-Beta to enhance the epoxide selectivity
in the liquid-phase epoxidation of alkenes.
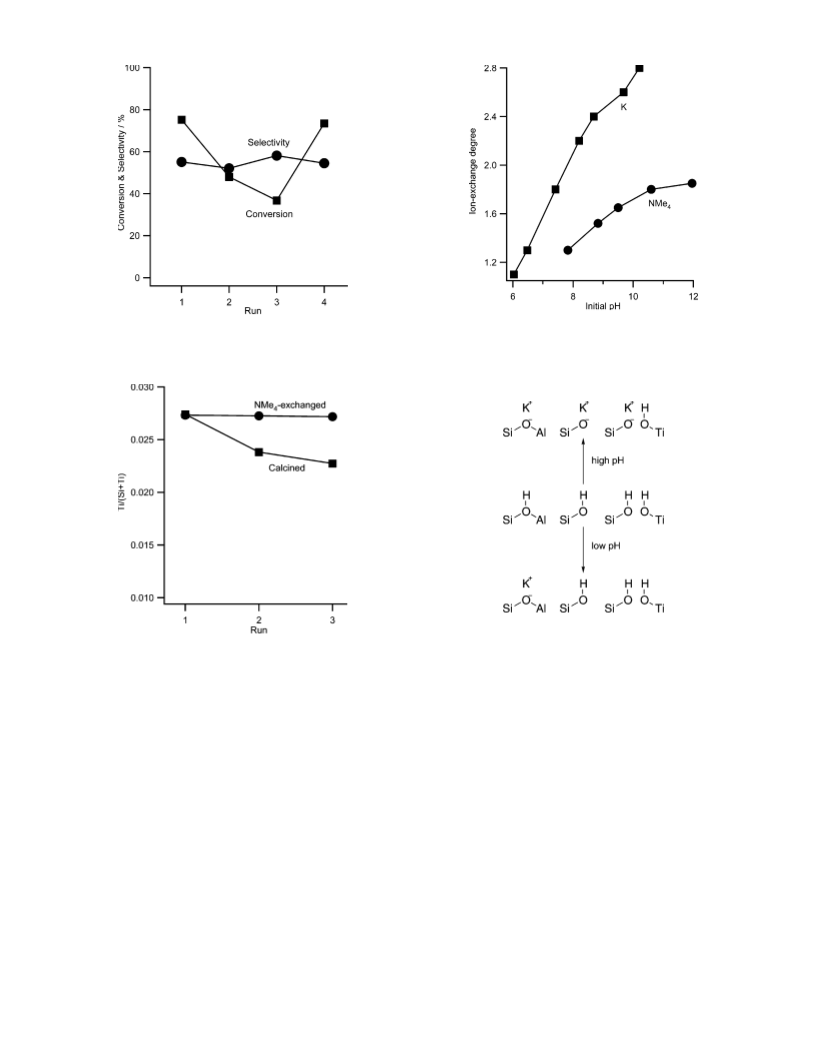
cation of the ion-exchange reagent and the initial pH in the ion
exchange. In all the cases, the final pH values of the ion-
exchange ranged between 6 and 7.
1
2.2. Characterizations. Powder X-ray diffraction patterns
were collected on a MAC Science MX-Labo diffractometer with
an accelerating voltage and a tube current of 40 kV and 20 mA,
respectively. Elemental analyses were performed on a Shimadzu
ICPS-8000E photoemission spectrometer equipped with induc-
tively coupled plasma for inorganic elements and on a Perkin-
Elmer 2400 series II CHNS/O analyzer for organic elements.
FT-IR spectra were collected on a Perkin-Elmer 1600 FT-IR
spectrometer using a KBr pellet technique (a 2 mg sample was
diluted with 100 mg of KBr and shaped into a 10 mm o.d. pellet)
or a self-supporting technique (30 mg sample shaped into a 20
21
29
27
mm o.d. pellet). Si- and Al-MAS NMR spectroscopies were
performed on a JEOL ECA-400 multinuclear solid-state mag-
netic resonance spectrometer at a magnetic field of 79.6 and
104 MHz, respectively. Nitrogen adsorption measurements were
carried out on a BEL Japan BELSORP 28SA automatic gas
adsorption instrument. The sample was preevacuated under a
pressure of 1.33 Pa at 473 K for 3 h.
2.3. Epoxidation of Alkenes. Epoxidation of alkenes was
carried out batchwise in a water-bathed 20 mL round-bottom
flask equipped with a Dimroth condenser and a magnetic stirrer;
2
. Experimental Section
.1. Catalyst Preparation. [Ti,Al]-Beta was hydrothermally
5
mL of methanol or acetonitrile as a solvent, 8.25 mmol of
cyclohexene, 1-hexene, or 2-hexenes (cis/trans isomers ratio )
1/59), and 2.5 mmol of H2O2 (31 wt % in water) and catalyst
50 mg for cyclohexene and 100 mg for 1-hexene) were mixed
2
5,6
synthesized according to the literature by using tetraethylam-
monium hydroxide (TEAOH) as a structure-directing agent
4
(
(SDA) with a modification to decrease the formation of
and heated under vigorous stirring at 333 K. After the reaction,
the reactor was quenched in an ice bath and the reaction mixture
was centrifuged to separate the liquid phase from the catalyst.
Quantitative analyses were performed on a Shimadzu GC-14B
gas chromatograph equipped with an OV-1 50 m capillary
column using cycloheptanone as the internal standard.
extraframework anatase. Titanium tetrabutoxide (TBOT, Kanto
Chemicals) was employed as a Ti source instead of ethoxide,
and TBOT was hydrolyzed in the aqueous H2O2 (31 wt % in
water, Mitsubishi Gas Chemicals) to stabilize mononuclear Ti
species of peroxotitanate. H2O2/Ti ratio was 10. This procedure
was performed in an ice bath. The resultant yellow solution of
peroxotitanate was added to the TEAOH solution (35 wt % in
water, Aldrich), and the mixture was continuously stirred for
2
.4. Ring-Opening Reaction of Epoxide. To clarify which
sites catalyze the solvolysis of epoxides, the solvolysis reactions
of cyclohexene oxide catalyzed by various zeolitic materials
with or without the addition of peroxide were examined. The
reactions were performed in a similar way to the alkene
epoxidation. TS-1 (Si/Ti ) 80) and three [Ti,Al]-Beta samples
1
.5 h. Precipitated silica (Nipsil VN-3, SiO2/Al2O3 ) 457, SiO2/
Na2O ) 227) and aluminum nitrate nonahydrate (Koso, Al-
NO3)3‚9H2O) were added to the above solution to give a mother
gel with a molar composition of SiO2‚0.033TiO2‚0.005Al2O3‚
.28TEA2O‚12.5H2O. After the gel was aged at room temper-
(
(cal[793], cal[793]-ie(NMe4)9.50 and cal[793]-ie(K)8.15), Sili-
0
calite-1, H-ZSM-5 (JRC-Z5-25H, Japan Reference Catalyst,
Si/Al ) 12.5), H-Beta (JRC-Z-HB25, Si/Al ) 12.5) and
deboronated B-Beta (Si/B > 500) were employed as catalysts.
ature for 2 h, hydrothermal synthesis was carried out in a PTFE-
lined stainless steel autoclave with agitation under an autogenous
pressure at 413 K for 4 days.
5
0 mg of catalyst, 5 mL of methanol as solvent and 2.5 mmol
SDA cations packing the channels of as-synthesized [Ti,Al]-
Beta were removed by the following two procedures: (a) as-
synthesized [Ti,Al]-Beta was calcined at T K in the O2 stream,
of cyclohexene oxide were heated to 333 K in the presence or
the absence of oxidant (2.5 mmol of H2O2 (31 wt % in water)
or tert-butyl hydroperoxide (TBHP; (CH3)3COOH, 70 wt % in
water)). Water was externally added when neither oxidant nor
TBHP was added, to adjust the amount of H2O to that present
in the H2O2 added.
where T was 473 and 793 K, which was designated as cal[T];
3
(
b) as-synthesized [Ti,Al]-Beta was treated with 6.0 mol dm-
aqueous ammonium nitrate solution twice to extract SDA,
followed by the calcination at T, where T was varied in the
range of 373-793 K, which was designated as ex(AN)-cal-
3
. Results and Discussion
[
793] (AN stands for ammonium nitrate). Acetic acid (1.0 mol
3.1. Effect of SDA-Extraction Treatment. Results of
-
3
dm ) and hydrochroric acid were also used to extract SDA.
After the treatment, the dried samples were calcined at 473 K
for 10 h. These samples were designated as ex(AA)-cal[473]
catalytic epoxidation of cyclohexene over directly calcined and
SDA-extracted [Ti,Al]-Beta are shown in Table 1. Directly
calcined [Ti,Al]-Beta (cal[793]) converted cyclohexene actively
but yielded no cyclohexene oxide after 3 h (run 1). The main
products were 1,2-cylohexanediol and its monomethyl ether due
to the acid-catalyzed solvolysis of cyclohexene oxide, together
with small amount of allylic oxidation products. The negligible
formation of cyclohexene oxide strongly indicates that there
were a considerable number of acid sites in just calcined [Ti,Al]-
Beta. The catalyst prepared by treating as-synthesized [Ti,Al]-
Beta with ammonium nitrate followed by mild calcination at
(AA stands for acetic acid) and ex(HCl)-cal[473], respectively.
Alternatively, [Ti,Al]-Beta, calcined [Ti,Al]-Beta, cal[793], was
-3
treated with 1.0 mol dm quaternary ammonium acetate (NR4-
OAc, R ) Me, Et, Pr), ammonium acetate (NH4OAc), or alkali
or alkaline earth metal acetate solution and successively calcined
at 473 K. The ion-exchange degrees were controlled by the pH
of the solution. Samples prepared by the ion exchange are
designated as cal[793]-ie(M)x, where M and x represents the

 Goa, Yasuhide
Goa, Yasuhide
 Wu, Peng
Wu, Peng
 Tatsumi, Takashi
Tatsumi, Takashi