THE OZONIZATION OF MODEL LIGNIN COMPOUNDS
1145
acid derivatives by highꢀperformance liquid chromaꢀ
tography.
D270
1.6
2
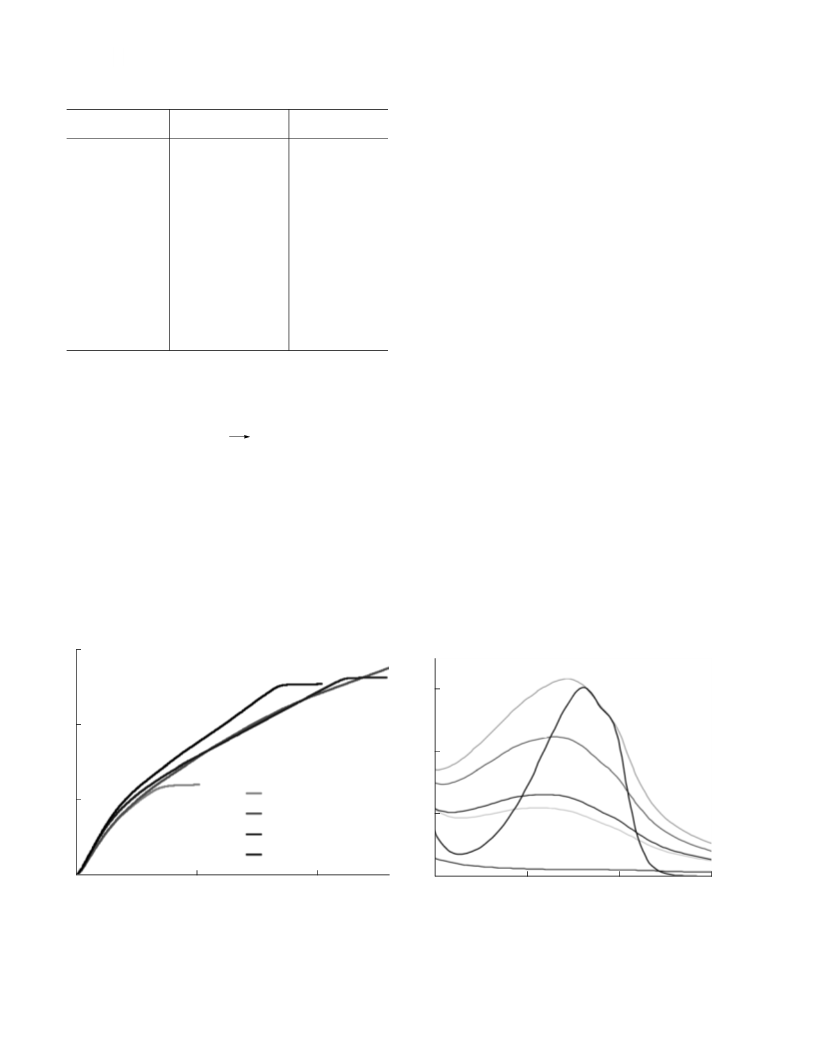
Similar curves were obtained in the ozonolysis of
1
veratrole catalyzed by Mn(II) ions. The spectral data
were used to construct the dependence of the optical
density at the maximum of veratrole absorption D270
on the time of ozonization in the presence of mangaꢀ
nese ions (Fig. 5). According to Fig. 5, Mn(II) ions do
not influence the destruction of the aromatic ring of
veratrole and muconic acid derivatives formed as a
result of ring opening under the action of ozone. The
ozonolysis of veratrole is a multistage process, and
ozone is spent for the cleavage of aromatic ring C–C
bonds and C–C bonds in intermediates such as
muconic and maleic acids and their derivatives, which
at high rates transform into lowꢀmolecularꢀweight
compounds, such as glyoxalic, oxalic, and formic
acids. It can therefore be suggested that an increase in
the consumption of ozone in the presence of Mn(II)
ions is related to the activity of metal ions in the oxidaꢀ
tion of saturated carboxylic acids.
1.2
0.8
0.4
0
250
500
750
t, s
Fig. 5. Dependences of optical density
D
4
on the time of
270
veratrole ozonization; [Ver] = 6
×
10⎯ M, [О ] = 1.3
–5
×
0
3 en
–3
10 M, and [Mn(II)] = (1) 0 and (2) 3.4 × 10 M.
Similar trends were observed for the reaction of
ozone with guaiacol (the phenol fragment of the lignin
macromolecule) in the presence of Mn(II) ions. At the
initial reaction stages, when the specific consumption
of ozone is 2.5 mol per mole of guaiacol, the rate of the
reaction does not change as metal concentration
increases. As with veratrole, Mn(II) ions do not influꢀ
ence guaiacol aromatic ring destruction and the
destruction of muconic acid derivatives formed in
ozonolysis, which is substantiated by the 13C NMR
and UV data [18].
The mechanism of the action of Mn(III) ions
formed under the action of Mnꢀperoxidase on lignin is
discussed in [7, 8]. According to this mechanism, a
chelate complex of Mn(III) with a lowꢀmolecularꢀ
weight acid attacks the lignin phenol ring and initiates
radical processes, which play an important role in the
destruction of the aromatic structure of lignin. Nonꢀ
phenol aromatic structures present in lignin are not
attacked by the Mn(III) chelate complex [7, 8].
An increase in the specific consumption of ozone
to 5 M and the rate of the reaction in the presence of
Mn(II) ions is likely related to the catalysis by metal
ions of transformations of intermediate products of
deeper guaiacol oxidation.
The data given above allow us to suggest a hypoꢀ
thetical scheme of the destruction of aromatic strucꢀ
tures of lignin model compounds. According to this
scheme, Mn(III) chelate structures formed in the oxiꢀ
dation of veratrole and guaiacol with ozone do not
participate in the destruction of the aromatic structure
of the compounds. Note that the rate constants for the
ozonization of veratrole and catalyst are close, but, as
distinct from guaiacol, veratrole does not contain the
OH group for interaction with Mn(III) chelate comꢀ
plexes. For guaiacol, the rate constant for ozonization
is higher by an order of magnitude than the rate conꢀ
stant for the oxidation of Mn(II) ions. Therefore, it
cannot be ruled out that Mn(III) chelate complexes
do not influence the destruction of the guaiacol aroꢀ
matic ring for kinetic reasons.
The experimental data allowed us to make certain
suggestions concerning the role played by Mn(II) in
the mechanism of catalytic transformations of lignin
model compounds under the action of ozone. Ozone,
which is a strong electrophilic agent, easily attacks
sites with an increased electron density, that is, aroꢀ
matic structures and compounds containing double
bonds. Simultaneously, ozone oxidizes Mn(II) ions.
As shown above, the oxidation of manganese ions
involves the stage of the formation of Mn(III) ions,
which are stabilized by lowꢀmolecularꢀweight prodꢀ
ucts of the oxidation of aromatic compounds (acids
and ketoacids) with the formation of chelate comꢀ
plexes. The redox potential and stability of Mn(III)
are determined by the type and concentration of the
organic acid that plays the role of the chelating agent.
These complexes are strong oxidants and can particiꢀ
pate in transformations of various organic comꢀ
pounds. Mn(III) then transforms into Mn(II) as a
result of intramolecular transfer.
To summarize, the mechanism of the action of
Mn(II) ions in the ozonocatalytic oxidation of phenol
and nonphenol lignin structural units does not contraꢀ
dict the mechanisms discussed in the literature for the
action of lignolytic enzymes on lignin and its model
compounds.
RUSSIAN JOURNAL OF PHYSICAL CHEMISTRY A
Vol. 84
No. 7
2010

 Mitrofanova
Mitrofanova
 Khudoshin
Khudoshin
 Lunin
Lunin