
Journal of Physical Chemistry p. 1360 - 1368 (1991)
Update date:2022-08-17
Topics:
 Gay, Ian D.
Gay, Ian D.
 McFarlan, A. J.
McFarlan, A. J.
 Morrow, B. A.
Morrow, B. A.
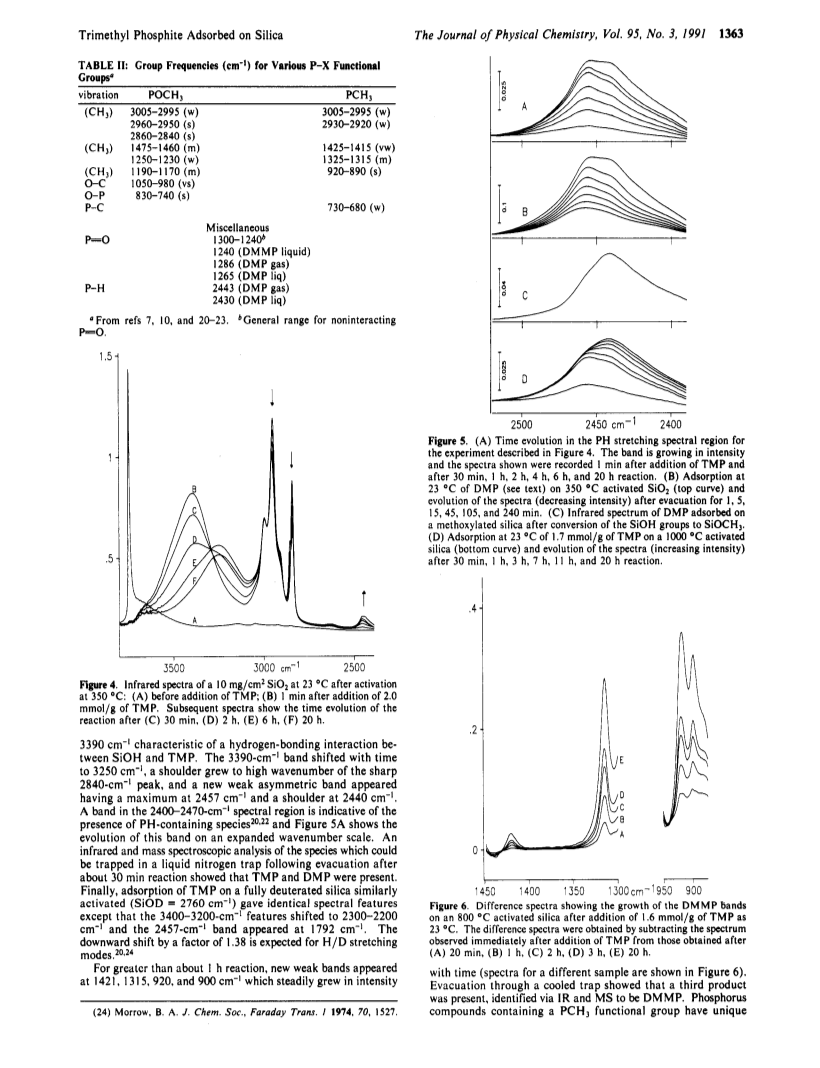
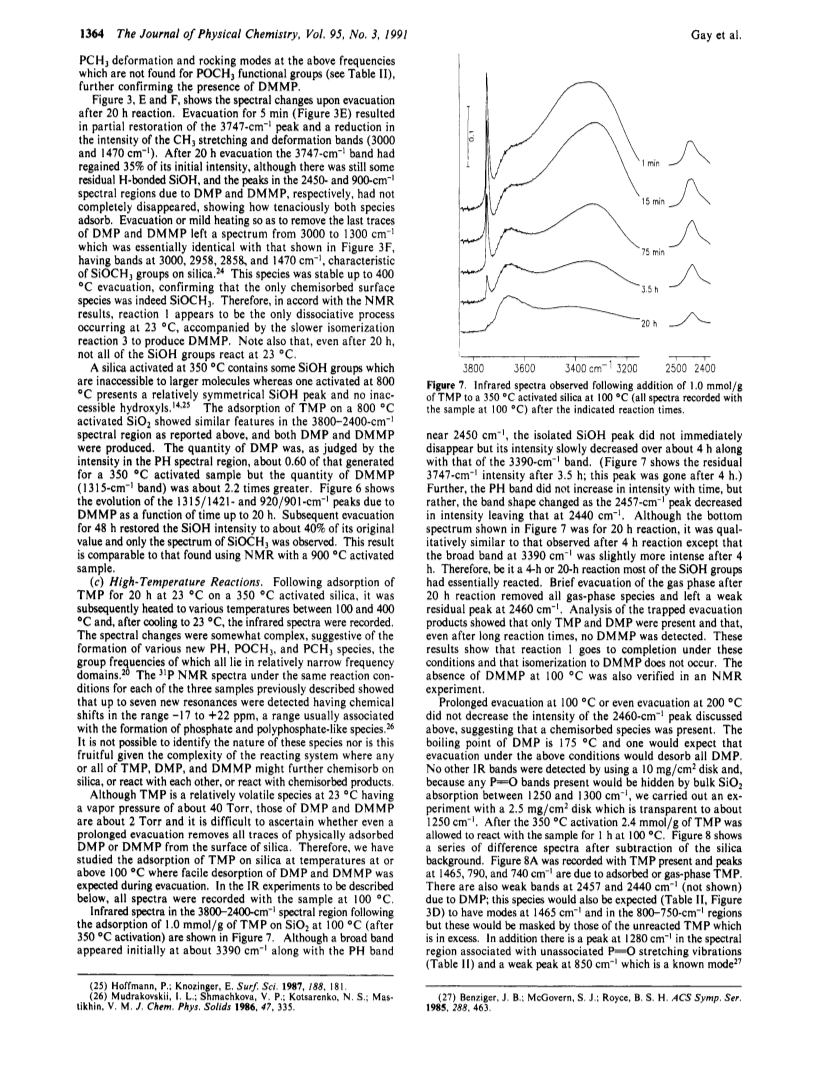
Infrared spectroscopy and phosphorus-31 magic angle spinning nuclear magnetic resonance spectroscopy have been used to study the adsorption of trimethyl phosphite (TMP) on silica.At 23 deg C TMP reacts rapidly with surface silanol groups to give SiOCH3 as a chemisorbed product and liquid dimethyl phosphite (DMP).However, formation of DMP ceases when about half of the SiOH groups have been consumed because DMP strongly hydrogen bonds to the remaining silanols thereby inhibiting further reaction between TMP and SiOH.TMP also undergoes isomerization to dimethyl methylphosphonate (DMMP) which is catalyzed by SiOH.As the number of initial silanol groups is decreased (by using higher temperatures of vacuum activation) the quantity of DMP produced decreases whereas that of DMMP increases.A mechanism for formation of DMP and DMMP has been suggested.At 100 deg C isomerization does not occur, all SiOH groups are consumed, and the major product is DMP/SiOCH3 accompanied by a small quantity of a chemisorbed phosphorus-containing species having the proposed structure (SiO)2P-H(=O).The latter is stable up to 400 deg C.If TMP is heated with silica from 100 to 400 deg C, in addition to SiOCH3, the major new chemisorbed product of the reaction which can be identified by IR and NMR is (SiO)2P-Me(=O) (Me = CH3).The advantages of a combined IR-NMR approach are discussed.
View More






Hangzhou Yanshan Chemical Co.,Ltd.
Contact:86-571- 87698076
Address:Room 1001, #1 Building, Zhongtian MCC, No.2 Youzhinong, Wenyi West Road, Xihu District, Hangzhou, China
Chengdu Gelipu Biotechnology Co., Ltd.
website:http://www.glp-china.com
Contact:86-28-82610909
Address:chegndu
ZHEJIANG JIANYE CHEMICAL CO.,LTD.
Contact:86-571-64149273,64149234
Address:No. 48, Fuxi Road, Meicheng Town
website:http://www.sagechem.com
Contact:+86-571-86818502
Address:Room C1301, New Youth Plaza, 8 Jia Shan Road, Hangzhou, China
Jiangsu Zhenfang Chemical CO.,LTD.(Suzhou Zhenfang Chemical Factory)
Contact:+86-512-69598882
Address:Room1201, Jiayuan Road No.1018, Xiangcheng District, Suzhou, China
Doi:10.1016/j.tet.2003.08.045
(2003)Doi:10.1007/BF02291538
(1997)Doi:10.1016/S0040-4039(00)95315-2
(1987)Doi:10.1016/j.tet.2006.03.050
(2006)Doi:10.1016/0040-4020(80)87018-9
(1980)Doi:10.1021/jm980671w
(1999)