Y.-F. Han, J.H. Lunsford / Journal of Catalysis 230 (2005) 313–316
315
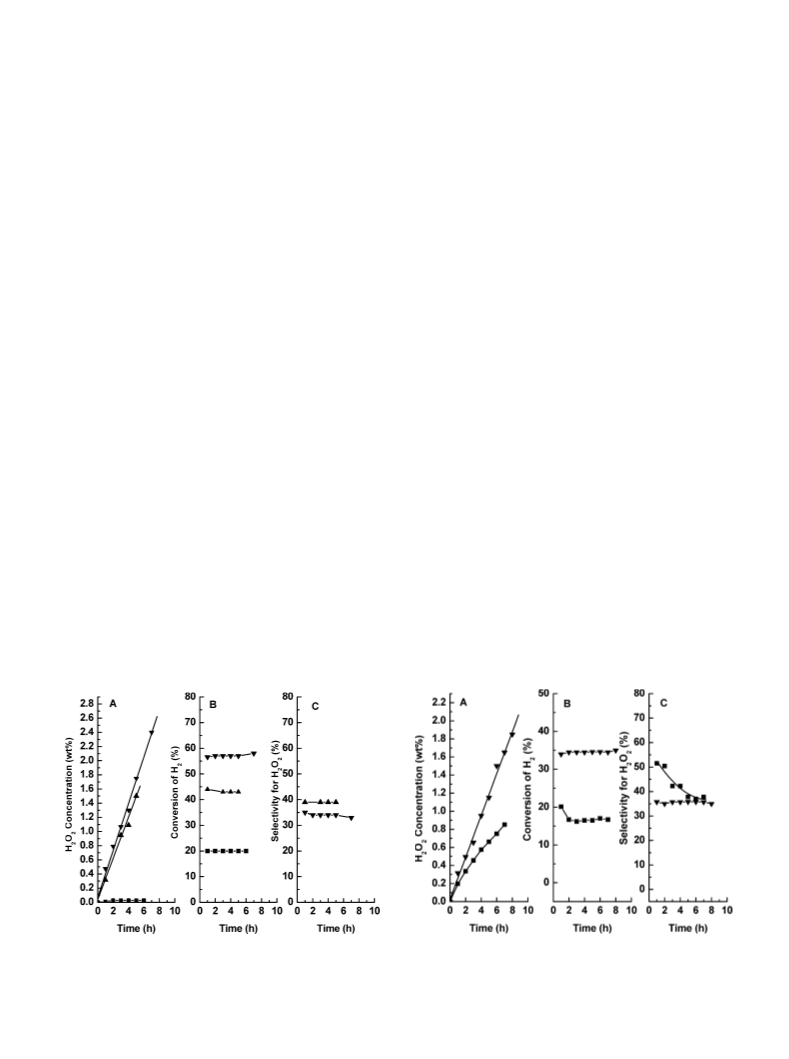
The selectivity decreased from 51 to 36% over 5 h. The
latter selectivity is almost the same as that obtained with
the HCl/ethanol system and is comparable to those obtained
in the H2SO4/ethanol systems. The similarities between the
ethanol and aqueous systems are somewhat fortuitous, how-
ever, since the state of the Pd varies between the two cases
as noted above. The variation of rate with time, as observed
for the aqueous system, is typical of the colloid, which is
intrinsically unstable.
In the previous study [7] it was found that in pure ethanol
(no acid added) a small amount of H2O2 appeared during
the first hour, but thereafter there was no net formation of
the peroxide. Similar results were also reported for an aque-
ous system [5]. Meanwhile, in ethanol the H2 conversion
was 45%, and when H2O2 was added to the system, it de-
composed over a period of 4 h, even in the presence of
the reagents. These results confirm that one of the roles
of the acid (protons) is to inhibit the subsequent reduction
of H2O2 by hydrogen, as has been pointed out previously
[6,7]. Other investigators have suggested that the role of the
acid is to prevent the base-catalyzed decomposition of H2O2
[1,8,10]; however, with ethanol as the liquid phase, this
seems unlikely. In a separate experiment H2O2 was added to
nonacidified ethanol that contained the catalyst and O2. Be-
fore the addition of H2O2, the system had been exposed to
both reagents. The H2O2 concentration (0.8 wt%) decreased
slightly at first and then remained constant for a period of
4 h; that is, in the absence of an acid the H2O2 did not de-
compose.
3.3. Combinations of HCl, H2SO4, water, and ethanol
In an effort to elucidate the roles of each component, sev-
eral combinations of the two acids and the two liquids were
studied; the results are presented in Fig. 3. In all cases 50 mg
of Pd/SiO2 was present. First, the reaction was carried out in
an equal volume mixture of ethanol and water with H2SO4
as the acid. After 7 h, 0.8 wt% H2O2 was formed, which is
about one-third of that produced in pure ethanol (Fig. 1A).
The H2 consumption was less than that in pure ethanol but
more than that in pure water. Here water seems to have a di-
lution effect, but it does not completely inhibit the reactions
when H2SO4 is the acid.
3.4. Secondary reactions in the H2SO4/ethanol system
Even in the presence of the acid, secondary reactions do
occur, and these result in the loss of selectivity. This effect is
illustrated by the results summarized in Table 1 and Fig. 4.
The amount of catalyst was increased from 20 to 120 mg,
and the concentration of H2O2, the conversion of H2, and
the selectivity were determined after 5 h on stream. Whereas
the H2 conversion continued to increase with catalyst load-
ing, the concentration of H2O2 reached a maximum at a
loading of 40 mg and decreased such that with 120 mg of
catalyst almost no H2O2 was detected. The peroxide selec-
tivity reached a maximum at a loading of 30 mg of catalyst.
The role of secondary reactions in these phenomena is
confirmed by the results in Fig. 4. In these experiments the
reaction was allowed to proceed for 2 h, and then ca. 0.8 wt%
H2O2 was added to the system. When 30 mg of catalyst was
present, the subsequent formation rate of H2O2 formation
remained the same; however, when 90 mg of catalyst was
added, the amount of H2O2 actually decreased with time.
These results may be understood in terms of consecutive
reactions in which H2O2 is first formed and then is subse-
A second experiment involved the addition of HCl to the
H2SO4/water system after the reaction had proceeded for
2 h. During the first 2 h the formation rate of H2O2 was
very small, as expected, but when 0.17 N HCl was added the
rate increased to about the level observed for the HCl/water
system (Fig. 2A). From this result one can conclude that sul-
fate ions do not poison the catalyst when water is the liquid
phase.
Table 1
Effect of catalyst amount on H O concentration, H conversion and selec-
2
2
2
a
tivity for H O
2
2
Amount of
catalyst (mg)
H O
concentration (%)
H conversion
2
(%)
Selectivity
for H O (%)
2
2
2
2
20
30
40
50
70
1.3
1.5
1.9
1.7
0.5
0.1
Trace
35
37
49
55
65
68
75
32
45
40
34
10
1.9
Fig. 3. Catalytic formation of H O in 0.24 N H SO /water (30 ml)–
2
2
2
4
ethanol (30 ml): (") concentration of H O and (!) conversion of H .
2
2
2
90
120
Catalytic formation of H O in 0.24 N H SO /water solution: (2) concen-
2
2
2
4
Trace
tration of H O and (1) H conversion. After 2 h, 0.24 N H SO /water
2
2
2
2
4
a
solution was made 0.17 N in HCl: (Q) concentration of H O .
5 wt% Pd/SiO , 0.24 N H SO /ethanol, after 5 h reaction.
2 2 4
2
2

 Han, Yi-Fan
Han, Yi-Fan
 Lunsford, Jack H.
Lunsford, Jack H.