10.1246/cl.1984.1931
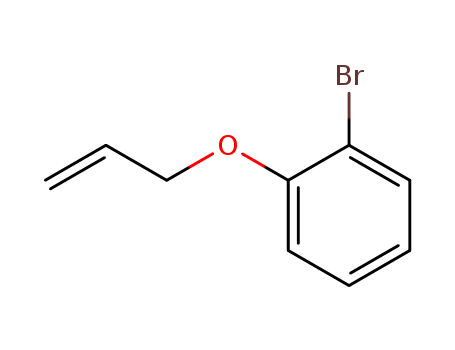
The study investigates the copper (I)-induced radical cyclization of diethyl o-(allyloxy)phenylmalonates to chroman derivatives. The key chemicals involved are o-(allyloxy)bromobenzenes and copper (I) diethyl malonate. The o-(allyloxy)bromobenzenes react with copper (I) diethyl malonate to form diethyl o-(allyloxy)phenylmalonates, which then undergo cyclization under the reaction conditions to produce various chroman derivatives such as 4,4-bis(ethoxycarbonyl)3-methylchromans, 4,4-bis(ethoxycarbonyl)-3-methylenechromans, and 3-(3-butenyl)-4,4-bis(ethoxycarbonyl)chromans. The copper (I) diethyl malonate plays a crucial role in this process, as the homolysis of the Cu(I)-C bond generates the radical species necessary for the cyclization to occur. The study also examines how different substituents on the aromatic ring and at the o-allyloxyl group influence the product distribution, finding that electron-withdrawing substituents accelerate the initial aromatic substitution reaction and lead to better isolation yields of chromans.