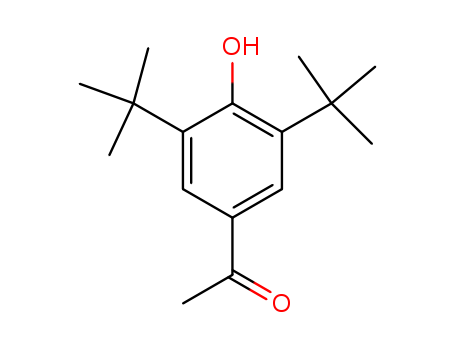
- Chemical Name:3,5-DI-TERT-BUTYL-4-HYDROXYACETOPHENONE
- CAS No.:14035-33-7
- Molecular Formula:C16H24 O2
- Molecular Weight:248.365
- Hs Code.:2914501900
- Mol file:14035-33-7.mol
Synonyms:Acetophenone, 3',5'-di-tert-butyl-4'-hydroxy-(6CI,7CI,8CI); 2,6-Di-tert-butyl-4-acetylphenol; 3,5-Di-tert-butyl-4-hydroxyphenylmethyl ketone;3',5'-Di-tert-butyl-4'-hydroxyacetophenone; 4-Acetyl-2,6-di-tert-butylphenol;4'-Hydroxy-3',5'-di-tert-butylacetophenone