10.1039/c39810000482
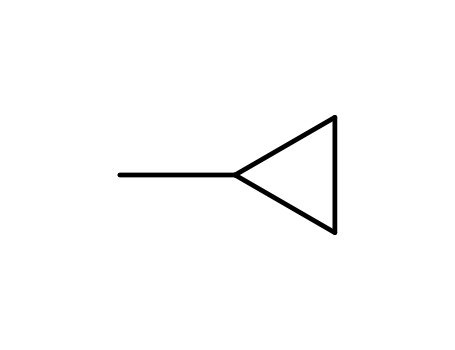
The research investigates the oxidation capabilities of the mono-oxygenase system from Methylococcus capsulatus on various substrates including cyclopropane, methylcyclopropane, and certain aromatic compounds. The study found that cyclopropane is oxidized to cyclopropanol, while methylcyclopropane yields cyclopropylmethanol. For aromatic substrates, the enzyme shows regioselectivity, oxidizing monosubstituted benzenes to para-substituted phenols and exhibiting an NIH shift in the oxidation of ethylbenzene. The oxidation products were identified using techniques such as gas-liquid chromatography (g.l.c.), thin-layer chromatography (t.l.c.), and nuclear magnetic resonance (n.m.r.) spectroscopy. The results suggest that the enzyme's mechanism involves the conversion of dioxygen into a metal-bound oxygen species capable of inserting into C-H bonds and adding to C=C bonds. The study also noted spatial restrictions at the enzyme's active site, as larger substituents like t-butyl and 1-methylcyclopropyl-benzene were not oxidized.