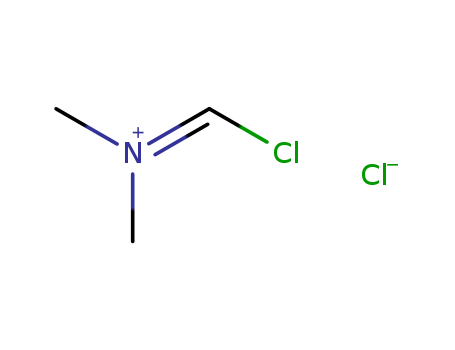
- Chemical Name:N-(Chloromethylidene)-N-methylmethanaminium chloride
- CAS No.:3724-43-4
- Deprecated CAS:1487-66-7,92458-33-8,1512534-40-5,92458-33-8
- Molecular Formula:C3H7ClN*Cl
- Molecular Weight:128.001
- Hs Code.:29252000
- European Community (EC) Number:425-970-6,609-368-2
- UNII:Q39V7G8776
- DSSTox Substance ID:DTXSID7044423
- Wikipedia:Vilsmeier_reagent
- Wikidata:Q27894258
- ChEMBL ID:CHEMBL3183181
- Mol file:3724-43-4.mol
Synonyms:3724-43-4;(Chloromethylene)dimethyliminium chloride;arnold's reagent;Vilsmeier reagent;N-(Chloromethylidene)-N-methylmethanaminium chloride;(CHLOROMETHYLENE)DIMETHYLAMMONIUM CHLORIDE;Methanaminium, N-(chloromethylene)-N-methyl-, chloride;MFCD00011868;N-(chloromethylene)-N-methylmethanaminium chloride;chloromethylene dimethylammonium chloride;chloromethylidene(dimethyl)azanium;chloride;DTXSID7044423;Dimethylchloromethyliminium chloride;N-Chloromethylene-N,N-dimethyl ammonium chloride;chloro-n,n-dimethylformiminium chloride;Chloromethylenedimethylammonium chloride;Q39V7G8776;N,N-Dimethylchloromethyliminium chloride;Chloromethylene(dimethyl)ammonium chloride;EC 425-970-6;EC 609-368-2;[chloromethylene]dimethylammonium chloride;(Chloromethylene)dimethylammonium chloride (6CI,7CI);Ammonium, (chloromethylene)dimethyl-, chloride (8CI);Viismeier reagent;Dimethylchloroformiminium chloride;(Chlormethylene)dimethylammonium chloride;C3H7ClN.Cl;SCHEMBL280;C3-H7-Cl-N.Cl;CHEMBL3183181;DTXCID5024423;UNII-Q39V7G8776;Tox21_302163;GEO-04109;AKOS017345161;chlormethylene dimethylammonium chloride;FS-4034;(chloromethylene)dimethylammoniumchloride;chloromethylene-dimethylammonium chloride;chloromethylene dimethyl-ammonium chloride;N,N,-Dimethylchloromethyliminium chloride;NCGC00255861-01;(Chloromethlene)dimethyl ammonium chloride;(chloromethylidene)dimethylazanium chloride;(chloro-methylene)dimethylammonium chloride;(chloromethylene)-dimethylammonium chloride;(Chloromethylene)-dimethylammonium-chloride;(Chloromethylene)dimethyl ammonium chloride;CAS-3724-43-4;(chloro-methylen)-dimethyl-ammonium-chloride;FT-0604890;D89427;Q27894258;Methanaminium, N-(chloromethylene)-N-methyl-, chloride (1:1)