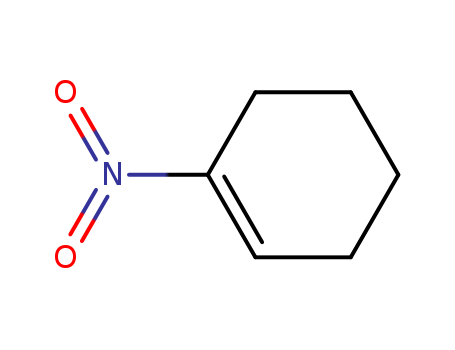
- Chemical Name:1-Nitrocyclohexene
- CAS No.:2562-37-0
- Molecular Formula:C6H9 N O2
- Molecular Weight:127.143
- Hs Code.:2904209090
- European Community (EC) Number:219-883-0
- UNII:H6PSH8C6BM
- DSSTox Substance ID:DTXSID40180294
- Nikkaji Number:J192.062F,J3.558.640G
- Wikidata:Q27144116
- Metabolomics Workbench ID:66642
- Mol file:2562-37-0.mol
Synonyms:1-Nitrocyclohexene;1-Nitro-1-cyclohexene;2562-37-0;1-Nitrocyclohex-1-ene;Cyclohexene, 1-nitro-;1-nitro-cyclohexene;H6PSH8C6BM;EINECS 219-883-0;(E)-1-Nitrocyclohexene;UNII-H6PSH8C6BM;SCHEMBL458470;1-Nitro-1-cyclohexene, 99%;CHEBI:73802;DTXSID40180294;BCP29176;AKOS015912668;AS-57236;CS-0204678;EN300-93754;J3.558.640G;F77997;Nitrocyclohexene, 1-;(1-Nitrocyclohex-1-ene);J-016070;Q27144116;NYH