10.3390/molecules23051093
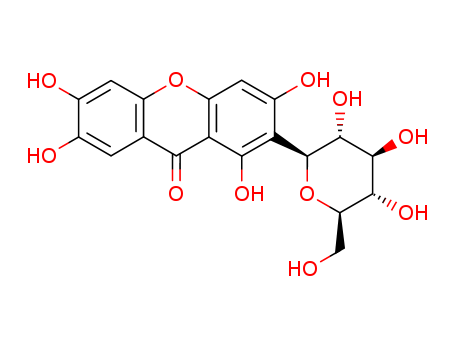
The research focuses on the synthesis and biological evaluation of new glycosylated flavonoids and xanthones with inhibitory activity on cell growth. The study involves the synthesis of four flavonoids (compounds 5, 6, 9, and 10) and one xanthone (compound 7) containing acetoglycoside moieties. Key chemicals used in the synthesis include rutin, diosmin, mangiferin, and 3,4-dihydroxyflavone as starting materials. Acetic anhydride and microwave irradiation were employed for acetylation, while 2,3,4,6-tetra-O-acetyl-α-D-glucopyranosyl bromide was used as the glucose donor for glycosylation. The synthesized compounds were evaluated for their in vitro cell growth inhibitory activity against six human tumor cell lines, including A375-C5, MCF-7, NCI-H460, U251, U373, and U87MG. The results showed that compound 10 exhibited the most potent inhibitory activity across all tested cell lines, with GI50 values < 8 μM and notable selectivity for cancer cells. The study highlights the importance of both acetylation and glycosylation in enhancing the biological activity of these compounds, providing insights for future development of more effective antitumor agents.


 T+,
T+, N
N



