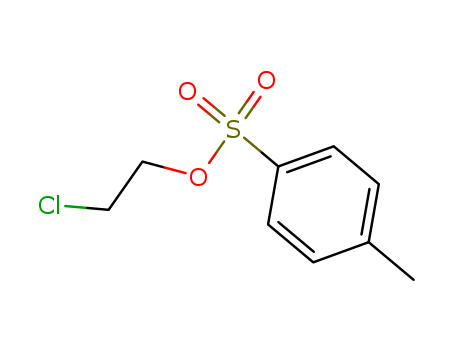
- Chemical Name:2-Chloroethyl p-toluenesulfonate
- CAS No.:80-41-1
- Deprecated CAS:56215-02-2,85426-03-5,344310-40-3
- Molecular Formula:C9H11 Cl O3 S
- Molecular Weight:234.704
- Hs Code.:29055900
- European Community (EC) Number:201-277-2
- NSC Number:6079
- DSSTox Substance ID:DTXSID20883256
- Nikkaji Number:J48.050I
- Wikidata:Q72450136
- ChEMBL ID:CHEMBL166116
- Mol file:80-41-1.mol
Synonyms:80-41-1;2-Chloroethyl p-toluenesulfonate;2-Chloroethyl 4-methylbenzenesulfonate;2-Chloroethyl tosylate;2-Chloroethyl p-tosylate;2-Tosyloxyethyl chloride;Ethanol, 2-chloro-, 4-methylbenzenesulfonate;2-Chloroethanol p-toluenesulfonate;p-Toluenesulfonic acid, 2-chloroethyl ester;2-CHLOROETHYLP-TOLUENESULFONATE;2-(p-Toluenesulfonyloxy)ethyl chloride;NSC 6079;Ethanol, 2-chloro-, p-toluenesulfonate;2-chloroethyl-p-toluenesulfonate;p-Toluenesulfonic acid-2-chloroethyl ester;beta-Chloroethyl p-toluenesulfonate;2-Chloroethyl toluene-4-sulphonate;EINECS 201-277-2;2-Chloroethanol 4-methylbenzenesulfonate;BRN 1968816;AI3-02279;2-Chlorethylester kyseliny p-toluensulfonove;2-Chloroethyl para-toluenesulfonate;Ethanol, 2-chloro-, 1-(4-methylbenzenesulfonate);.beta.-Chloroethyl p-toluenesulfonate;2-Chlorethylester kyseliny p-toluensulfonove [Czech];MFCD00000970;p-TOLUENESULFONIC ACID, beta-CHLOROETHYL ESTER;beta-Chloroethylester kyseliny p-toluensulfonove [Czech];beta-Chloroethylester kyseliny p-toluensulfonove;p-Toluenesulfonic acid, .beta.-chloroethyl ester;.beta.-Chloroethylester kyseliny p-toluensulfonove;4-11-00-00249 (Beilstein Handbook Reference);p-Toluenesulfonic Acid 2-Chloroethyl Ester;chloroethyltosylate;chloroethyl tosylate;chloroethyl-tosylate;2-chloroethanol tosylate;beta-CHLOROETHYL-p-TOLUENE SULFONATE;2-chloro ethanol tosylate;1-chloro-2-tosyloxyethane;WLN: G2OSWR D1;2-chlorethyltoluen-4-sulfonat;SCHEMBL263231;2-chloroethyl-p-toluensulfonate;CHEMBL166116;2-chloroethyl p-toluensulphonate;2-chloroethyl p-toluenesulphonate;2-chloroethyl-p-toluenesulphonate;ZXNMIUJDTOMBPV-UHFFFAOYSA-;2-chloroethyl-p-toluene sulfonate;2-chloroethyl-p-toluene-sulfonate;2-chloroethyl-paratoluenesulfonate;DTXSID20883256;NSC6079;2-Chloroethyl p-toluene sulfonate;beta-chloroethyl p-toluenesulphonate;NSC-6079;1-chloro-2-p-toluenesulfonyloxyethane;AKOS005258286;(2-chloro-ethyl) toluene-4-sulphonate;2-Chloroethyl p-toluenesulfonate, 97%;2-CHLOROETHYL 4-TOLUENESULFONATE;2-Chloroethyl 4-methylbenzenesulfonate #;MS-20213;2-chloroethyl 4-methylbenzene-1-sulfonate;LS-154164;p-toluenesulfonic acid-beta-chloroethylester;toluene-4-sulfonic acid 2-chloroethyl ester;C2380;CS-0130585;FT-0619529;Toluene-4-sulfonic acid 2-chloro-ethyl ester;D71096;4-methyl-benzenesulfonic acid 2-chloro-ethyl ester;A839904


 Xn
Xn




