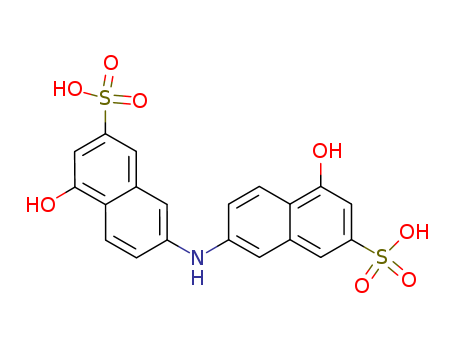
- Chemical Name:Rhoduline Acid
- CAS No.:87-03-6
- Molecular Formula:C20H15NO8S2
- Molecular Weight:461.473
- Hs Code.:2934999090
- European Community (EC) Number:201-719-4
- NSC Number:37203,1698
- UNII:EB8FE6GXT2
- DSSTox Substance ID:DTXSID8058952
- Nikkaji Number:J4.269B
- Wikidata:Q72506673
- ChEMBL ID:CHEMBL1093490
- Mol file:87-03-6.mol
Synonyms:87-03-6;Rhoduline Acid;I Acid Imide;J Acid Imide;Di-J acid;RW acid;7,7'-Iminobis(4-hydroxy-2-naphthalenesulfonic acid);NSC-1698;4,4'-Dihydroxy-7,7'-iminodi(naphthalene-2-sulphonic acid);EB8FE6GXT2;2-Naphthalenesulfonic acid, 7,7'-iminobis[4-hydroxy-;NSC 1698;EINECS 201-719-4;2-Naphthalenesulfonic acid, 7,7'-iminobis(4-hydroxy-;7,7'-azanediylbis(4-hydroxynaphthalene-2-sulfonic acid);6,6'-Iminobis(1-hydroxy-3-sulfonaphthalene);4-hydroxy-7-[(5-hydroxy-7-sulfonaphthalen-2-yl)amino]naphthalene-2-sulfonic acid;6,6'-Iminobis[1-hydroxy-3-sulfonaphthalene];2-Naphthalenesulfonic acid,7'-iminobis[4-hydroxy-;C20H15NO8S2;UNII-EB8FE6GXT2;SCHEMBL9236771;CHEMBL1093490;DTXSID8058952;NSC1698;NSC37203;4,4'-DIHYDROXY-7,7'-IMINODI(NAPHTHALENE-2-SULFONIC ACID);MFCD00035713;NSC-37203;AKOS040744485;FT-0609306;4,4'-dihydroxy-7,7'-iminodi(naphthalen-2-sulfonsyre);5,5-Dihydroxy-2,2-dinaphthylamine-7,7-disulphonicacid;2-NAPHTHALENESULPHONIC ACID, 7,7'-IMINOBIS(4-HYDROXY-;7,7'-IMINOBIS(4-HYDROXY-2-NAPHTHALENESULPHONIC ACID)