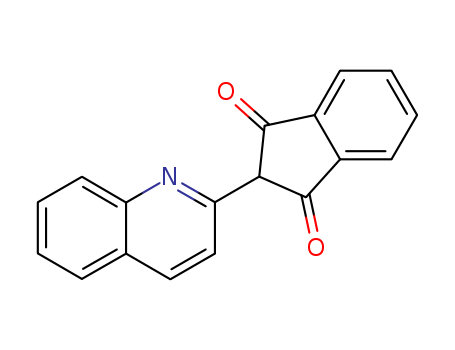
- Chemical Name:Quinoline Yellow
- CAS No.:83-08-9
- Molecular Formula:C18H11NO2
- Molecular Weight:273.291
- Hs Code.:2933499090
- European Community (EC) Number:305-897-5,201-453-9
- NSC Number:18950,7503,3047
- UNII:44F3HYL954
- DSSTox Substance ID:DTXSID00873130
- Nikkaji Number:J29.108K
- Wikipedia:Quinoline_Yellow_SS
- Wikidata:Q7272330
- NCI Thesaurus Code:C71670
- RXCUI:1305581
- Metabolomics Workbench ID:58717
- ChEMBL ID:CHEMBL1486465
- Mol file:83-08-9.mol
Synonyms:Quinoline Yellow;Quinophthalone;8003-22-3;Solvent yellow 33;11641 Yellow;Erio Chinoline Yellow 4G;2-quinolin-2-ylindene-1,3-dione;83-08-9;2-(2-Quinolyl)-1,3-indandione;D&C Yellow No. 11;Quinoline Yellow 2SF;2-(2-Quinolyl)-1,3-indanedione;Quinoline Yellow SS;2-(2-Quinolyl)-1H-indene-1,3(2H)-dione;D and C Yellow No. 11;D & C Yellow no. 11;D&C Yellow No 11;D & C Yellow 11;Arlosol Yellow S;Waxoline Yellow T;2-(2-Quinolinyl)-1H-indene-1,3(2H)-dione;Solvant Yellow 33;2-(quinolin-2-yl)-1H-indene-1,3(2H)-dione;D.C. Yellow Dye No. 11;Chinoline Yellow ZSS;Quinoline Yellow Base;Yellow 204;CHEBI:53700;Nitro Fast Yellow SL;D&C Yellow 11;C.I. SOLVENT YELLOW 33;D and C Yellow 11;NSC 18950;1H-Indene-1,3(2H)-dione, 2-(2-quinolinyl)-;CI 47000;CCRIS 1448;C.I. 47000;EINECS 201-453-9;EINECS 232-318-2;MFCD00011593;2-quinolin-2-yl-1H-indene-1,3(2H)-dione;1H-Indene-1, 2-(2-quinolinyl)-;C.I. 4700;Chinophthalon;QuinolineYellow;Amarelo Quinolina;quinoline yellow a;Quinoline Yellow (C.I. 47000);quinol ine yellow ss;Yellow 11;Quinophthalone (VAN);D&C Yellow No.11;DSSTox_CID_4906;Epitope ID:116214;DSSTox_RID_77574;Quinoline Yellow, 95.0%;DC YELLOW NO. 11;DSSTox_GSID_24906;SCHEMBL42716;YELLOW 11 [INCI];C.I. Solvent Y ellow 33;MLS000037978;UNII-44F3HYL954;KI204 [INCI];CHEMBL1486465;BDBM32227;cid_5355280;HSDB 8052;DTXSID00873130;NSC3047;NSC7503;2-(2-Quinolyl)indan-1,3-dione;44F3HYL954;CI 47000 [INCI];HMS2417K18;KI204;SOLVENT YELLOW 33 [INCI];NSC-3047;NSC-7503;NSC18950;2-(1,3-dioxoindan-2-yl)quinoline;2-(2-quinolyl)indane-1,3-quinone;Tox21_202504;BBL027785;NSC-18950;STK367449;Quinoline Yellow, Dye content 95 %;AKOS000282972;Solvent Yellow 33 / Quinoline Yellow;CCG-103550;LS-1801;NCGC00020851-01;NCGC00020851-02;NCGC00020851-03;NCGC00020851-04;NCGC00020851-05;NCGC00020851-06;NCGC00020851-07;NCGC00020851-08;NCGC00020851-09;NCGC00020851-10;NCGC00021766-02;NCGC00021766-03;NCGC00021766-04;NCGC00021766-05;NCGC00260053-01;AS-13999;Quinoline Yellow 2SF, analytical standard;SMR000035456;CAS-8003-22-3;2-(2-quinolyl)-1H-inden-1,3(2H)-dion;FT-0674285;2-(1H-quinolin-2-ylidene)indene-1,3-dione;2-(1H-quinolin-2-ylidene)indane-1,3-quinone;F87556;SR-01000002715;Q7272330;SR-01000002715-2;W-109300