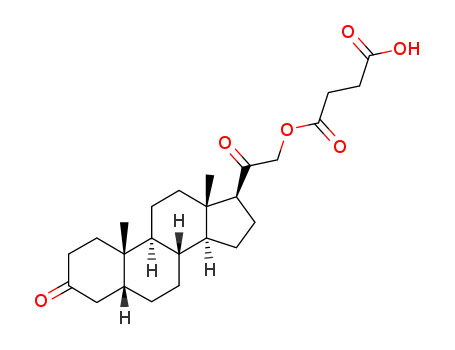
- Chemical Name:Hydroxydione hydrogen succinate
- CAS No.:80-96-6
- Molecular Formula:C25H36 O6
- Molecular Weight:432.557
- Hs Code.:
- European Community (EC) Number:201-314-2
- UNII:6LJJ67GNMV
- DSSTox Substance ID:DTXSID00861577
- Nikkaji Number:J192.580F
- Wikidata:Q27265106
- Metabolomics Workbench ID:199124
- ChEMBL ID:CHEMBL2110850
- Mol file:80-96-6.mol
Synonyms:Hydroxydione hydrogen succinate;80-96-6;Hydroxydionhydrogensuccinat;UNII-6LJJ67GNMV;6LJJ67GNMV;21-Hydroxy-5beta-pregnane-3,20-dione 21-(hydrogen succinate);EINECS 201-314-2;3,20-Dioxo-5beta-pregnan-21-yl hydrogensuccinat;5.beta.-Pregnane-3,20-dione, 21-hydroxy-, hydrogen succinate;Pregnane-3,20-dione, 21-(3-carboxy-1-oxopropoxy)-, (5.beta.)-;Hydroxydione Succinic Acid;21-hydroxy-5beta-pregnan-3,20-dion-21-(hydrogensuccinat);Pregnane-3,20-dione, 21-(3-carboxy-1-oxopropoxy)-, (5beta)-;CHEMBL2110850;DTXSID00861577;4-[2-[(5R,8R,9S,10S,13S,14S,17S)-10,13-dimethyl-3-oxo-1,2,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydrocyclopenta[a]phenanthren-17-yl]-2-oxoethoxy]-4-oxobutanoic acid;5beta-Pregnane-3,20-dione, 21-hydroxy-, succinate;Q27265106;21-HYDROXY-5.BETA.-PREGNANE-3,20-DIONE SUCCINATE;5.BETA.-PREGNANE-3,20-DIONE, 21-HYDROXY-, SUCCINATE;3-[(3,20-Dioxo-5beta-pregnan-21-yl)oxycarbonyl]propanoic acid;4-(2-((5R,8R,9S,10S,13S,14S,17S)-10,13-DIMETHYL-3-OXO-1,2,4,5,6,7,8,9,11,12,14,15,16,17-TETRADECAHYDROCYCLOPENTA(A)PHENANTHREN-17-YL)-2-OXO-ETHOXY)-4-OXO-BUTANOIC ACID