10.1021/je60055a035
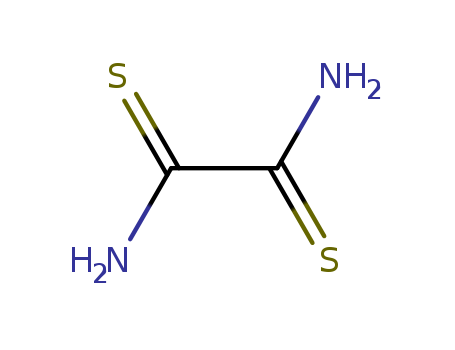
The study focuses on the synthesis and spectral data of certain chemical compounds. The first part of the study, conducted by Frank L. Setliff and Gary O. Rankin, involves the preparation of 2-chloro- and 2-bromo-5-fluoronicotinic acid and 6-chloro- and 6-bromo-5-fluoronicotinic acid through the oxidation of corresponding dihalo-3-picolines. The dihalo-3-picolines serve as precursors for the acids. The study also includes the preparation of amines Ie and If through dissolving-metal reduction, and the subsequent diazotization and thermal decomposition to produce other compounds. The second part of the study, conducted by George Y. Sarkis and Subhi Al-Azawe, involves the synthesis of nine bithiazole derivatives by the interaction of rubeanic acid with various α-haloketones. The study provides experimental and spectral data for these compounds. The chemicals involved, such as nicotinic acid, dihalo-3-picolines, rubeanic acid, and α-haloketones, play crucial roles in the synthesis processes and the formation of the target compounds.


 Xn
Xn




