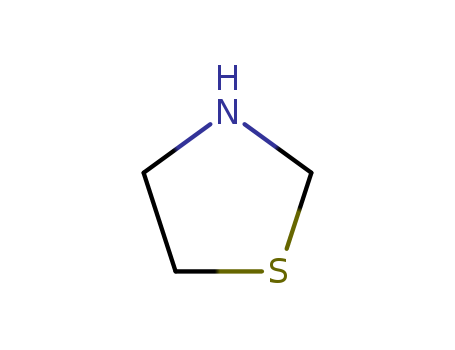
- Chemical Name:Thiazolidine
- CAS No.:504-78-9
- Molecular Formula:C3H7NS
- Molecular Weight:89.1613
- Hs Code.:29341000
- European Community (EC) Number:208-002-5
- NSC Number:357911
- UNII:I320806BKW
- DSSTox Substance ID:DTXSID10198447
- Nikkaji Number:J149.955F
- Wikipedia:Thiazolidine
- Wikidata:Q413761
- Metabolomics Workbench ID:57336
- ChEMBL ID:CHEMBL1916078
- Mol file:504-78-9.mol
Synonyms:Thiazolidine;Thiazolidines


 C
C




