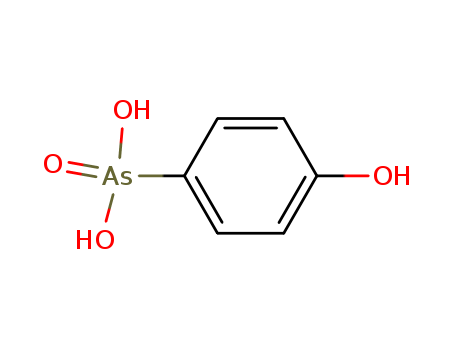
- Chemical Name:Oxarsanilic acid
- CAS No.:98-14-6
- Molecular Formula:C6H7 As O4
- Molecular Weight:218.041
- Hs Code.:2836400000
- European Community (EC) Number:202-641-3
- NSC Number:1943
- UNII:L49V552V77
- DSSTox Substance ID:DTXSID00243359
- Nikkaji Number:J54.495G
- Wikidata:Q27282689
- Mol file:98-14-6.mol
Synonyms:4-Hydroxyphenylarsonic acid;98-14-6;Oxarsanilic acid;(4-Hydroxyphenyl)arsonic acid;4-Hydroxybenzenearsonic acid;Arsonic acid, (4-hydroxyphenyl)-;p-Hydroxybenzenearsonic acid;Phenol-p-arsonic acid;(p-Hydroxyphenyl)arsonic acid;Phenol-para-arsonic acid;Benzenearsonic acid, p-hydroxy-;para-Hydroxyphenylarsonic acid;p-Hydroxyphenylarsonic acid;NSC 1943;EINECS 202-641-3;BRN 2936785;UNII-L49V552V77;AI3-14867;NSC-1943;L49V552V77;DTXSID00243359;4-16-00-01187 (Beilstein Handbook Reference);C6H7AsO4;C6-H7-As-O4;SCHEMBL3056859;WLN: Q-AS-QO & R DQ;DTXCID50165850;NSC1943;1-decyl-1-oxido-piperidin-1-ium;MFCD00045707;AKOS015856390;LS-29094;H0201;D90814;A845819;Q27282689