Refernces
10.1007/s00706-011-0563-x
The research focuses on the development of an eco-friendly and efficient catalytic system for the synthesis of a series of new (E)-1-(benzylideneamino)-3-cyano-6-(trifluoromethyl)-1H-2-pyridones. These compounds are derived from the cyclocondensation reaction of benzylidene cyanoacetohydrazide with 4-alkoxy-1,1,1-trifluoro-3-alken-2-ones, using a novel catalytic system consisting of triethylamine and a Lewis acid in the ionic liquid [BMIM][BF4]. The study emphasizes the comparison between this new system and traditional methods, highlighting that the products were only successfully synthesized using the new catalyst system. The experiments were conducted at room temperature, yielding products in moderate to good yields (42–87%). Various analytical techniques were employed to characterize the products, including NMR spectroscopy, GC-MS, and in some cases, X-ray diffraction, with elemental analysis and melting points also reported. The research underscores the potential of ionic liquids as green solvents, capable of being recycled and reused, thus reducing environmental impact.
10.1080/10426500802625594
The research aimed to synthesize new heterocyclic Schiff bases derived from 2-amino-3-formylchromone and investigate their antimicrobial activity. The study focused on combining chromone moieties with 1,2,4-triazole or 1,2,4-triazine derivatives through an azomethine linkage to create novel nitrogen heterocyclic systems. Key chemicals used included 2-amino-3-formylchromone as the starting material, various hydrazine derivatives (such as benzoylhydrazine, cyanoacetohydrazide, and thiocarbohydrazide) for condensation reactions, and electrophilic reagents like benzoyl chloride, acetic anhydride, and carbon disulfide for further heterocyclization. The newly synthesized compounds were characterized using IR, 1H NMR, and mass spectrometry. The antimicrobial activity of these compounds was tested against a range of bacteria and fungi, revealing that compounds 8 and 20 showed moderate activity against bacteria and high activity against fungi, while compounds 9, 13, and 15 exhibited high antifungal activity. The study concluded that the synthesized compounds have potential as antimicrobial agents, particularly those incorporating dithioxo-1,2,4-triazole and antipyrine moieties, and suggested that further modifications could enhance their biological efficacy.
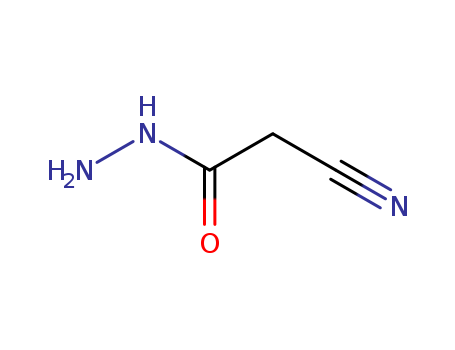
10.1016/j.cclet.2013.11.026
The research focuses on the synthesis of benzimidazole-bearing 2-pyridone derivatives as potential antimicrobial agents to combat multi-drug resistance in bacteria and fungi. The study employed molecular hybridization to combine the bioactive properties of 2-pyridones and benzimidazoles into a single molecular framework. The synthesis involved the preparation of intermediate compounds through reactions with cyanoacetic acid hydrazide and Knoevenagel products, followed by condensation with aromatic aldehydes in boiling ethanol. The synthesized compounds were characterized using elemental analysis, infrared (IR) spectroscopy, nuclear magnetic resonance (NMR) spectroscopy, and mass spectrometry. The antimicrobial activity of the compounds was evaluated in vitro using the broth dilution method against various bacterial and fungal strains, with chloramphenicol and ketoconazole as standard drugs. The study also assessed the cytotoxic effects of the most potent compounds on human cancer cell lines. The findings indicated that compounds with electron-withdrawing groups, particularly at the meta or para position of the phenyl ring, exhibited the highest antibacterial activity, while a chlorine-substituted compound showed the most potent antifungal activity, with none of the tested compounds showing significant cytotoxic effects.


 Xn
Xn



