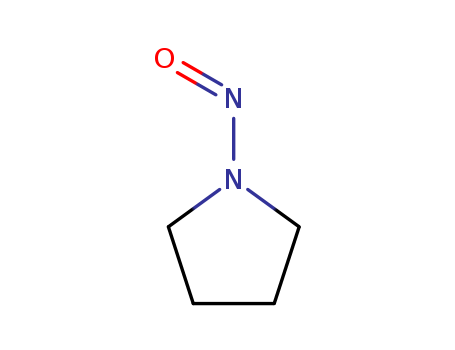
Chemical Property of N-Nitrosopyrrolidine
Chemical Property:
- Appearance/Colour:Light-Yellow Oil
- Vapor Pressure:0.233mmHg at 25°C
- Refractive Index:n20/D 1.489(lit.)
- Boiling Point:214°Cat760mmHg
- PKA:-3.14±0.20(Predicted)
- Flash Point:83.3°C
- PSA:32.67000
- Density:1.24g/cm3
- LogP:0.70150
- Storage Temp.:Refrigerator
- Solubility.:Chloroform (Sparingly), Ethyl Acetate, Methanol (Slightly)
- XLogP3:-0.2
- Hydrogen Bond Donor Count:0
- Hydrogen Bond Acceptor Count:3
- Rotatable Bond Count:0
- Exact Mass:100.063662883
- Heavy Atom Count:7
- Complexity:68.1
- Transport DOT Label:Poison
- Purity/Quality:
-
98%,99%, *data from raw suppliers
NPY1R *data from reagent suppliers
Safty Information:
- Pictogram(s):

- Hazard Codes:Xn
- Statements:
40
- MSDS Files:
-
SDS file from LookChem
Useful:
- Chemical Classes:Nitrogen Compounds -> Nitrosamines
- Canonical SMILES:C1CCN(C1)N=O
-
Description
N-Nitrosopyrrolidine (NPYR) is a nitrosamine compound that
is used primarily as a research chemical and is not produced
commercially. There is the possibility of generating NPYR after
frying in dry-cured bacon. The major amine precursors to NPYR
in cooked bacon are free proline in the adipose tissue and to
a lesser extent, collagenous connective tissues. When proline
peptides were heated with nitrite at pH 3.4, small amounts of
NPYR were formed from all tested peptides. There are several
potential sources of exposure of man to this group of potent
carcinogens. Certain foods were thought to be derived from the
interaction of nitrite with secondary amines in the food, either
spontaneously or by the agency of bacteria. Recently the
possibility that nitrosamines may be formed from secondary
amines and nitrite in the gastrointestinal tract has been
explored. The acid conditions that prevail in the human
stomach favor the nitrosation of dimethylamine. In addition,
at neutral pH, many secondary amines can be nitrosated by
nitrite or nitrate in the presence of intestinal bacteria, or the
cecal contents of the rat, and soluble enzymes which catalyze
the N-nitrosation of several secondary amines have been
extracted from two microorganisms. The rate of nitrosamine
formation depends greatly on the basicity of the amine; the less
basic amines such as diphenylamine and pyrrolidine are
nitrosated far more readily than the strongly basic dimethylamine
and diethylamine.
-
Uses
One of the N-nitroso compounds (NOCs) implicated in human colon carcinogenesis, but the toxicological mechanisms involved have not been elucidated. 1-Nitrosopyrrolidine is one of the N-nitroso compounds (NOCs) implicated in human colon carcinogenesis (1,2,3) but the toxicological mechanisms involved have not been elucidated. Drinking water contaminant candidate list 3 (CCL 3) compound as per United States Environmental Protection Agency (EPA), environmental, and food contaminants.