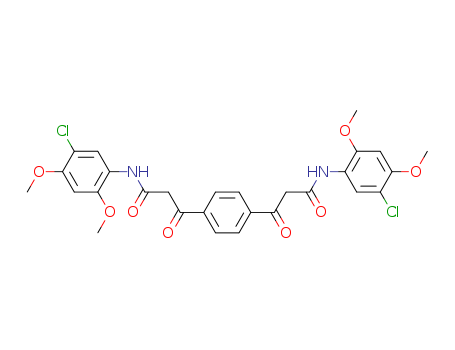
- Chemical Name:5,5'-Dichloro-2,2',4,4'-tetramethoxy-alpha,alpha'-terephthaloyldiacetanilide
- CAS No.:92-21-7
- Molecular Formula:C28H26 Cl2 N2 O8
- Molecular Weight:589.429
- Hs Code.:2924299090
- European Community (EC) Number:202-136-8
- DSSTox Substance ID:DTXSID20889334
- Nikkaji Number:J73.276A
- Wikidata:Q82869185
- Mol file:92-21-7.mol
Synonyms:92-21-7;A,A'-Terephthaloylbis-5-chloro-2,4-dimethoxyacetanilide;5,5'-Dichloro-2,2',4,4'-tetramethoxy-alpha,alpha'-terephthaloyldiacetanilide;EINECS 202-136-8;3-[4-[3-(5-chloro-2,4-dimethoxyanilino)-3-oxopropanoyl]phenyl]-N-(5-chloro-2,4-dimethoxyphenyl)-3-oxopropanamide;ALPHA,ALPHA-TEREPHTHALOYL-BIS-5-CHLORO-2,4-DIMETHOXYACETANILIDE;1,4-Benzenedipropanamide, N,N'-bis(5-chloro-2,4-dimethoxyphenyl)-.beta.,.beta.'-dioxo-;1,4-Benzenedipropanamide, N,N'-bis(5-chloro-2,4-dimethoxyphenyl)-beta,beta'-dioxo-;1,4-Benzenedipropanamide, N1,N4-bis(5-chloro-2,4-dimethoxyphenyl)-beta1,beta4-dioxo-;C28H26Cl2N2O8;DTXSID20889334;C28-H26-Cl2-N2-O8;AKOS024420947;C.I.37615;FT-0755502;A,A-TEREPHTHALOYLBIS-5-CHLORO-2,4-DIMETHOXYACETANILIDE;5,5'-dichlor-2,2',4,4'-tetramethoxy-alpha-alpha'-terephthaloyldiacetanilid;ALPHA,ALPHA'-TEREPHTHALOYL-BIS[5-CHLORO-2,4-DIMETHOXYACETANILIDE];1,4-Benzenedipropanamide, N1,N4-bis(5-chloro-2,4-dimethoxyphenyl)-.beta.1,.beta.4-dioxo-;3,3'-(1,4-phenylene)bis(N-(5-chloro-2,4-dimethoxyphenyl)-3-oxopropanamide)