- Chemical Name:Slaked lime
- CAS No.:1305-62-0
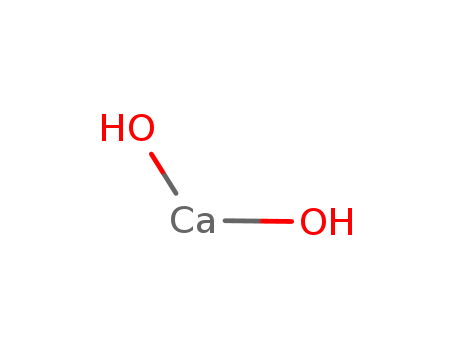
- Molecular Formula:Ca(OH)2
- Molecular Weight:74.0927
- Hs Code.:2825 90 19
- European Community (EC) Number:215-137-3
- UNII:PF5DZW74VN
- Nikkaji Number:J43.728J
- Wikidata:Q182849
- ChEMBL ID:CHEMBL2106388
- Mol file:1305-62-0.mol
Synonyms:Slaked lime;Hydrated lime;Lime hydrate;Lime water;Calcium hydroxide (Ca(OH)2);Slaked-lime;Calcarea caustica;Ca(OH)2;INS NO.526;CHEBI:31341;Calcium Oxide, Hydrated;E-526;calcium(II) hydroxide;Kalziumhydroxid;Loeschkalk;Kalsiumhydroksid;geloeschter Kalk;Hydroxyde de calcium;calcium(2+) hydroxide;Calcium hydroxide (ACN;CAH (CHRIS Code);CaH2O2;Calcium Hydroxide USP/FCC;LIME WATER [VANDF];Calcium hydroxide ACS grade;CALCIUM HYDROXIDE [II];CALCIUM HYDROXIDE [MI];Calcium hydroxide - Total dust;CALCIUM HYDROXIDE [FCC];CALCIUM HYDROXIDE [JAN];CALCAREA CAUSTICA [HPUS];CALCIUM HYDROXIDE [HSDB];CALCIUM HYDROXIDE [INCI];CHEMBL2106388;CALCIUM HYDROXIDE [VANDF];INS-526;AXCZMVOFGPJBDE-UHFFFAOYSA-L;CALCIUM HYDROXIDE [MART.];CALCIUM HYDROXIDE [USP-RS];CALCIUM HYDROXIDE [WHO-DD];AKOS015903965;Calcium hydroxide - Respirable fraction;CALCIUM HYDROXIDE [EP MONOGRAPH];CALCIUM HYDROXIDE [USP MONOGRAPH];FT-0623399;Q182849;J-005828


 Xi,
Xi, C
C




