10.1016/j.bmcl.2009.02.097
The research aimed to investigate the Gi protein-activating properties of a series of synthetic compounds by measuring their induced binding of [35S]GTPcS to different subtypes of Gi protein. The study encountered a significant issue due to the strong affinity between [35S]GTPcS and the tested compounds, which are characterized by multiple positive charges and high lipophilicity. The conclusion was that caution must be exercised when conducting binding studies with charged, highly lipophilic molecules. The chemicals used in the process included 4-aminopiperidines, 4,4’-bipiperidines, piperazines, and 4-methylalkylaminopiperidines, among others, which were synthesized using various reagents and conditions as detailed in the article's schemes and supplementary data.


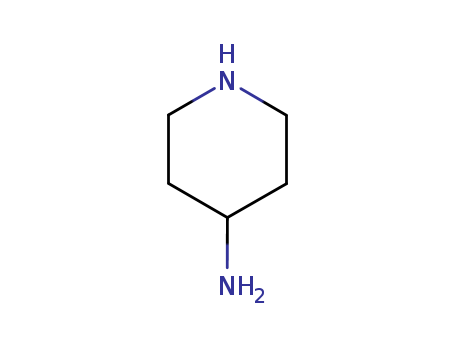
 C,
C, Xi
Xi




