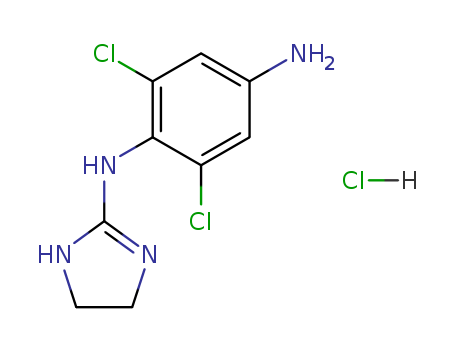
- Chemical Name:P-AMINOCLONIDINE
- CAS No.:73217-88-6
- Molecular Formula:C9H10Cl2N4*ClH
- Molecular Weight:281.572
- Hs Code.:
- Mol file:73217-88-6.mol
Synonyms:1,4-Benzenediamine,2,6-dichloro-N1-(4,5-dihydro-1H-imidazol-2-yl)-, dihydrochloride (9CI);Apraclonidine dihydrochloride; p-Aminoclonidine dihydrochloride