10.1021/ja5023909
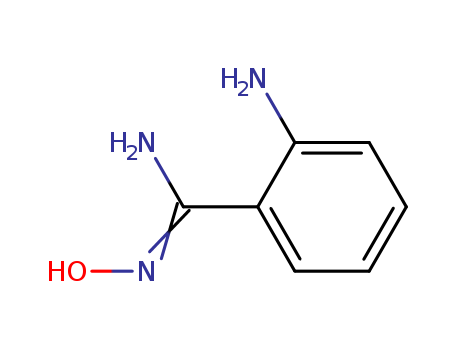
The research focuses on the rapid and hydrolytically stable modification of aldehyde-terminated proteins and phage libraries using 2-amino benzamidoxime (ABAO) derivatives. The purpose of this study was to develop a method for the unnatural modification of proteins, cell labeling, synthesis of antibody-drug conjugates, and other applications by reacting ABAO with aldehydes in water. The researchers found that the reaction between ABAO and aldehydes is kinetically similar to oxime formations performed under stoichiometric aniline catalysis, with the rate-determining step being the formation of a Schiff base, followed by rapid intramolecular ring closure. The reaction was accelerated by substituents in the aromatic ring that increase the basicity of the aromatic amine. The most reactive derivative identified was para-methoxy ABAO (PMA), which showed a rate of up to 40 M?1 s?1 with electron-rich aldehydes at pH 4.5. The product of the reaction, a dihydroquinazoline derivative, was found to be hydrolytically stable and showed fluorescence at 490 nm, suggesting the potential for developing fluorogenic aldehyde-reactive probes based on the ABAO framework. The study concludes that the ABAO framework offers a platform for new bioconjugation strategies, fluorogenic probes, and post-translational diversification of genetically-encoded libraries, with the added benefit of changes in both absorbance and fluorescence spectra during the reaction, facilitating rapid and accurate kinetic measurements on complex substrates.