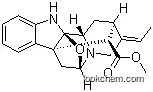
4684-32-6 Usage
Description
An Alstonia alkaloid obtained from A. scholaris R. Br., this base also occurs in
Rauwolfia vornitora. It has [α]D - 47° (CHCI3). The ultraviolet spectrum in
neutral solution (EtOH) has absorption maxima at 237 and 287 mil, while in
acidic solution (perchloric acid), there are absorption maxima at 239, 244 and
305 mil. A methoxycarbonyl group, an alkyl group and an ether bridge are present
in the molecule. The picrate is obtained as light yellow crystals with m.p. 172-
4°C (dec.) and the methiodide has m.p. 235-7°C.
Uses
Picrinine is a monoterpenoid indole alkaloid extracted from Ochrosia elliptica, displaing the enhancement of immunomodulatory activity and apaptosis inducing in the A549 cell lines.
References
Preparation:
Britten, Smith.,J. Chern. Soc., 3850 (1963)
Isolation:
Chatterjee et al., Tetrahedron Lett., 3633 (1965)
Check Digit Verification of cas no
The CAS Registry Mumber 4684-32-6 includes 7 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 4 digits, 4,6,8 and 4 respectively; the second part has 2 digits, 3 and 2 respectively.
Calculate Digit Verification of CAS Registry Number 4684-32:
(6*4)+(5*6)+(4*8)+(3*4)+(2*3)+(1*2)=106
106 % 10 = 6
So 4684-32-6 is a valid CAS Registry Number.
InChI:InChI=1/C20H22N2O3/c1-3-11-10-22-15-8-12(11)17(18(23)24-2)19-9-16(22)25-20(15,19)21-14-7-5-4-6-13(14)19/h3-7,12,15-17,21H,8-10H2,1-2H3/b11-3-/t12-,15-,16-,17?,19-,20-/m0/s1
4684-32-6Relevant articles and documents
Total Synthesis of (-)-Picrinine, (-)-Scholarisine C, and (+)-5-β-Methoxyaspidophylline
Zou, Peng,Yang, Hongjian,Wei, Jian,Wang, Taimin,Zhai, Hongbin
, p. 6836 - 6840 (2021)
The first asymmetric total synthesis of three picrinine-type akuammiline alkaloids, (-)-picrinine, (-)-scholarisine C, and (+)-5-β-methoxyaspidophylline, has been accomplished. The synthesis features an efficient acid-promoted oxo-bridge ring-opening and further carbonyl O-cyclization to assemble the furoindoline scaffold, an unusual Dauben-Michno oxidation to construct the requisite α,β-unsaturated aldehyde functionality, and a nickel-mediated reductive Heck reaction to forge the [3.3.1]-azabicyclic core.