Products Categories
| CAS No.: | 120-61-6 |
|---|---|
| Name: | DIMETHYL TEREPHTHALATE |
| Article Data: | 225 |
| Molecular Structure: | |
|
|
|
| Formula: | C10H10O4 |
| Molecular Weight: | 194.187 |
| Synonyms: | 1,4-Benzenedicarboxylicacid, dimethyl ester (9CI);Terephthalic acid, dimethyl ester (6CI,7CI,8CI);DMT;Dimethyl 1,4-benzenedicarboxylate;Dimethyl p-benzenedicarboxylate;Dimethyl p-phthalate;Methyl 4-(carbomethoxy)benzoate;Methyl p-(methoxycarbonyl)benzoate;NSC 3503; |
| EINECS: | 204-411-8 |
| Density: | 1.175 g/cm3 |
| Melting Point: | 140 °C |
| Boiling Point: | 285 °C at 760 mmHg |
| Flash Point: | 148 °C |
| Solubility: | insoluble in water |
| Appearance: | White needle crystal |
| Risk Codes: | 36/37/38 |
| Safety: | 24/25 |
| Transport Information: | UN 3256 |
| PSA: | 52.60000 |
| LogP: | 1.25980 |
- 144851-82-1METHYL2-AMINO-3-FLUOROBENZOATE
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 1219080-61-1IMIDAZOLE-2-BORONIC ACID

| Conditions | Yield |
|---|---|
| Stage #1: methanol; terephthalic acid for 0.5h; Reflux; Inert atmosphere; Stage #2: With thionyl chloride for 10h; Reflux; Inert atmosphere; | 100% |
| With thionyl chloride Heating; | 99% |
| Stage #1: methanol; terephthalic acid for 0.5h; Reflux; Stage #2: With thionyl chloride for 12h; Reflux; | 99% |

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane | 100% |
- 67-56-1
methanol

- 68-36-0
1,4-bis(trichloromethyl)benzene

- 120-61-6
1,4-benzenedicarboxylic acid dimethyl ester

| Conditions | Yield |
|---|---|
| at 20℃; for 6h; UV-irradiation; | 99% |
| With zinc(II) chloride |

| Conditions | Yield |
|---|---|
| With titanium silicate; dihydrogen peroxide for 8h; Heating; | 99% |
| With tert.-butylhydroperoxide In decane at 25℃; for 6h; Inert atmosphere; | 88% |
| With tert.-butylhydroperoxide In decane at 25℃; for 8h; Inert atmosphere; | 86% |

| Conditions | Yield |
|---|---|
| With 3-mesityl-4-methylthiazol-3-ium bis((trifluoromethyl)sulfonyl)imide; dimethyl sulfoxide; 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran Mechanism; Concentration; Electrochemical reaction; Inert atmosphere; Flow reactor; | 99% |
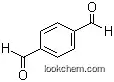
| Stage #1: methanol With 1,8-diazabicyclo[5.4.0]undec-7-ene; 1,4-dimethyl-1,2,4-triazolium iodide In tetrahydrofuran for 0.0833333h; Inert atmosphere; Stage #2: methyl 4-formylbenzoate With 3,5,3',5'-tetra-tert-butyl-4,4'-diphenoquinone In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; | 97% |
| With 2,2':6,2''-terpyridine; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; sodium acetate; sodium hydroxide at 90℃; for 12h; Green chemistry; chemoselective reaction; | 92% |
- 67-56-1
methanol

- 201230-82-2
carbon monoxide

- 619-44-3
methyl 4-iodobenzoate

- 120-61-6
1,4-benzenedicarboxylic acid dimethyl ester

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 100℃; under 5171.62 Torr; for 6h; Autoclave; | 99% |
| With palladium/palladium oxide-supported N-doped carbon at 120℃; under 15001.5 Torr; for 24h; Autoclave; Sealed tube; | 55% |
| palladium diacetate In N,N-dimethyl-formamide under 760 Torr; for 0.5h; Ambient temperature; |

- 83-48-7Stigmasta-5,22-dien-3-ol,(3b,22E)-
- 496-72-03,4-Diaminotoluene
- 18742-02-41,3-Dioxolane,2-(2-bromoethyl)-
- 106685-41-0Mehtyl 6-[3-(1-adamanty)-4-methoxy phenyl]-2-naphthoate
- 108-87-2Cyclohexane, methyl-
- 130-95-0Cinchonan-9-ol, 6'-methoxy-, (8alpha,9R)-
- 30674-80-72-Isocyanatoethyl methacrylate
- 93703-24-38-Bromo-3-methyl-xanthine
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin

What can I do for you?
Get Best Price
Consensus Reports
Specification
The IUPAC name of Dimethyl terephthalate is dimethyl benzene-1,4-dicarboxylate. With the CAS registry number 120-61-6, it is also named as Methyl p-(methoxycarbonyl)benzoate. The product's categories are Color Former & Related Compounds; Functional Materials; Sensitizer; Alpha Sort; Pesticides & Metabolites; C10 to C11; Carbonyl Compounds; Esters. It is white needle crystal which is insoluble in water, soluble in methanol, ether, acetone and chloroform. What's more, it is stable and incompatible with strong acids, strong bases, strong oxidizing agents. When heated to decomposition it emits acrid smoke and irritating fumes. Additionally, this chemical should be sealed in the container and stored in the cool and ventilate place.
The other characteristics of this product can be summarized as: (1)ACD/LogP: 2.47; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.47; (4)ACD/LogD (pH 7.4): 2.47; (5)ACD/BCF (pH 5.5): 44.66; (6)ACD/BCF (pH 7.4): 44.66; (7)ACD/KOC (pH 5.5): 528.01; (8)ACD/KOC (pH 7.4): 528.01; (9)#H bond acceptors: 4; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 4; (12)Index of Refraction: 1.514; (13)Molar Refractivity: 49.79 cm3; (14)Molar Volume: 165.2 cm3; (15)Polarizability: 19.74×10-24 cm3; (16)Surface Tension: 40.5 dyne/cm; (17)Enthalpy of Vaporization: 52.4 kJ/mol; (18)Vapour Pressure: 0.00288 mmHg at 25°C; (19)Rotatable Bond Count: 4; (20)Exact Mass: 194.057909; (21)MonoIsotopic Mass: 194.057909; (22)Topological Polar Surface Area: 52.6; (23)Heavy Atom Count: 14; (24)Complexity: 192.
Preparation of Dimethyl terephthalate: It is obtained by oxidation of p-xylene and subsequent esterification with methyl alcohol.
CH3C6H4CH3 + [O] → HOOCC6H4COOH
HOOCC6H4COOH + 2CH3OH → CH3OOCC6H4COOCH3
Uses of Dimethyl terephthalate: It is used in the production of polyesters, including polyethylene terephthalate and polytrimethylene terephthalate. It is also used in the synthesis of polyester fibers, resins, films, polyester paint and engineering plastics. In addition, it can react with phenylmagnesium bromide to get 1,4-bis-(a-hydroxy-benzhydryl)-benzene. The yield is 50%.
.gif)
When you are using this chemical, please be cautious about it as the following:
It is flammable by fire, heat and oxidants. It is also irritating to eyes, respiratory system and skin. So people should avoid contact with skin and eyes.
People can use the following data to convert to the molecule structure.
1. Smiles:c1(C(OC)=O)ccc(C(OC)=O)cc1
2. InChI:InChI=1/C10H10O4/c1-13-9(11)7-3-5-8(6-4-7)10(12)14-2/h3-6H,1-2H3
The following are the toxicity data which has been tested.
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | skin | > 5gm/kg (5000mg/kg) | "Industrial Hygiene and Toxicology," 2nd ed., Patty, F.A., ed., New York, John Wiley & Sons, Inc., 1958-63Vol. 2, Pg. 1906, 1963. | |
| rat | LD50 | intraperitoneal | 3900mg/kg (3900mg/kg) | BEHAVIORAL: MUSCLE WEAKNESS | American Industrial Hygiene Association Journal. Vol. 34, Pg. 455, 1973. |
| rat | LD50 | oral | > 3200mg/kg (3200mg/kg) | "Industrial Hygiene and Toxicology," 2nd ed., Patty, F.A., ed., New York, John Wiley & Sons, Inc., 1958-63Vol. 2, Pg. 1906, 1963. |