Products Categories
| CAS No.: | 143-08-8 |
|---|---|
| Name: | 1-Nonanol |
| Article Data: | 236 |
| Molecular Structure: | |
|
|
|
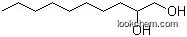
| Formula: | C9H20O |
| Molecular Weight: | 144.257 |
| Synonyms: | Nonyl alcohol(8CI);1-Hydroxynonane;NSC 5521;Octyl carbinol;Pelargonic alcohol;n-Nonan-1-ol;n-Nonyl alcohol;n-Nonanol; |
| EINECS: | 205-583-7 |
| Density: | 0.826 g/cm3 |
| Melting Point: | ?8-?6°C(lit.) |
| Boiling Point: | 211.617 °C at 760 mmHg |
| Flash Point: | 97.778 °C |
| Solubility: | 1 g/L (20 ºC) |
| Appearance: | colourless to light yellow liquid |
| Hazard Symbols: |
 Xn, Xn,  N N
|
| Risk Codes: | 20-51/53 |
| Safety: | 23-24/25-61-29 |
| Transport Information: | UN 3082 9/PG 3 |
| PSA: | 20.23000 |
| LogP: | 2.72930 |
- 144851-82-1METHYL2-AMINO-3-FLUOROBENZOATE
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE

| Conditions | Yield |
|---|---|
| With tri-n-butyl-tin hydride In methanol; diethyl ether for 4h; Reduction; Heating; | 100% |
| With hydrogenchloride; tetrahydrogenoboratebis(triphenylphosphine)copper(I) In dichloromethane | 94% |
| With hydrogenchloride; samarium In tetrahydrofuran at 20℃; | 94% |

| Conditions | Yield |
|---|---|
| With Methyltrichlorosilane; sodium iodide In acetonitrile at 25℃; for 9h; | A n/a B 100% |

| Conditions | Yield |
|---|---|
| With Methyltrichlorosilane; sodium iodide In acetonitrile at 25℃; for 9h; | 100% |

| Conditions | Yield |
|---|---|
| With [ReOCl2(1,2-bis(diphenylphosphino)ethane)]; hydrogen; potassium tetraphenylborate In toluene at 150℃; under 30003 Torr; for 30h; Autoclave; Inert atmosphere; | 99% |
| With hydrogen In hexane at 250℃; under 30003 Torr; for 3h; | 96.9% |
| With sodium tetrahydroborate; titanium tetrachloride In 1,2-dimethoxyethane for 14h; Ambient temperature; | 93% |
- 88773-83-5
decyloxy-tetrahydro-2H-pyran

- 143-08-8
nonyl alcohol

| Conditions | Yield |
|---|---|
| Nafion-H In methanol for 5h; | 98% |
| With magnesium bromide In diethyl ether for 4h; Ambient temperature; | 95% |
- 13038-21-6
non-8-en-1-ol

- 143-08-8
nonyl alcohol

| Conditions | Yield |
|---|---|
| With formic acid; tri-tert-butyl phosphine; palladium diacetate In tetrahydrofuran at 20℃; for 12h; | 97% |
| With formic acid; tri-tert-butyl phosphine; palladium diacetate In tetrahydrofuran at 20 - 60℃; for 12h; | 97% |
- 71733-81-8
tert-Butyl-dimethyl-nonyloxy-silane

- 143-08-8
nonyl alcohol

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In dichloromethane at 20℃; for 0.0833333h; atmospheric pressure; | 96% |

| Conditions | Yield |
|---|---|
| With zinc borohydride; silica gel In 1,2-dimethoxyethane for 0.5h; Ambient temperature; | 95% |
| With zinc borohydride; silica gel In 1,2-dimethoxyethane for 0.5h; Ambient temperature; other alkenes: regioselective anti-Markovnikov hydration; | 95% |
| With zinc borohydride; silica gel In N,N-dimethyl-formamide for 0.5h; Ambient temperature; | 75% |

- 143-08-8
nonyl alcohol

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In dichloromethane at 20℃; for 7h; atmospheric pressure; | 94% |
- 79137-09-0
1-Methoxymethoxy-nonane

- 143-08-8
nonyl alcohol

| Conditions | Yield |
|---|---|
| With magnesium bromide In diethyl ether for 36h; Ambient temperature; | 93% |
Specification
This chemical is called 1-Nonanol, and its systematic name is nonan-1-ol. With the molecular formula of C9H20O, its molecular weight is . The CAS registry number of this chemical is 143-08-8, and its product categories are 1-Alkanols; Monofunctional & Alpha,Omega-Bifunctional Alkanes; Monofunctional Alkanes. In additon, this chemical is colorless to slightly yellow liquid with a citrus odor similar to citronella oil, and it should be stored sealed in the cool and dry plcace.
Other characteristics of the 1-Nonanol can be summarised as followings: (1)ACD/LogP: 3.39; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 3.386; (4)ACD/LogD (pH 7.4): 3.386; (5)ACD/BCF (pH 5.5): 220.465; (6)ACD/BCF (pH 7.4): 220.465; (7)ACD/KOC (pH 5.5): 1655.651; (8)ACD/KOC (pH 7.4): 1655.651; (9)#H bond acceptors: 1; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 8; (12)Polar Surface Area: 20.23 Å2; (13)Index of Refraction: 1.432; (14)Molar Refractivity: 45.28 cm3; (15)Molar Volume: 174.602 cm3; (16)Polarizability: 17.95×10-24cm3; (17)Surface Tension: 29.509 dyne/cm; (18)Density: 0.826 g/cm3; (19)Flash Point: 97.778 °C; (20)Enthalpy of Vaporization: 52.098 kJ/mol; (21)Boiling Point: 211.617 °C at 760 mmHg; (22)Vapour Pressure: 0.041 mmHg at 25°C.
Production method of this chemical: The 1-Nonanol could be obtained by the reactant of nonanal. This reaction needs the reagent of Bu3SnH, and the solvents of methanol and diethyl ether. The yield is 100 %. In addition, this reaction should be taken for 4 hours, and the other condition is heating.
![]()
Uses of this chemical: The primary use of nonanol is in the manufacture of artificial lemon oil. Various esters of nonanol, such as nonyl acetate, are used in perfumery and flavors.
When you are using this chemical, please be cautious about it as the following: This chemical is harmful by inhalation. Do not breathe vapour. Avoid contacting with skin and eyes. It's also toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. Do not pull it into drains. Avoid releasing it to the environment. Refer to special instructions / safety data sheets when you use it.
You can still convert the following datas into molecular structure:
1.SMILES: CCCCCCCCCO
2.InChI: InChI=1/C9H20O/c1-2-3-4-5-6-7-8-9-10/h10H,2-9H2,1H3
3.InChIKey: ZWRUINPWMLAQRD-UHFFFAOYAC
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mammal (species unspecified) | LC50 | inhalation | 4360mg/m3 (4360mg/m3) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 51(5), Pg. 61, 1986. | |
| mammal (species unspecified) | LD50 | unreported | 19gm/kg (19000mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 51(5), Pg. 61, 1986. | |
| mouse | LC50 | inhalation | 5500mg/m3/2H (5500mg/m3) | "Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure," Izmerov, N.F., et al., Moscow, Centre of International Projects, GKNT, 1982Vol. -, Pg. 94, 1982. | |
| mouse | LD50 | intraperitoneal | 800mg/kg (800mg/kg) | "Patty's Industrial Hygiene and Toxicology," 3rd rev. ed., Clayton, G.D., and F.E. Clayton, eds., New York, John Wiley & Sons, Inc., 1978-82. Vol. 3 originally pub. in 1979; pub. as 2n rev. ed. in 1985.Vol. 2C, Pg. 4628, 1982. | |
| mouse | LD50 | oral | 6400mg/kg (6400mg/kg) | "Patty's Industrial Hygiene and Toxicology," 3rd rev. ed., Clayton, G.D., and F.E. Clayton, eds., New York, John Wiley & Sons, Inc., 1978-82. Vol. 3 originally pub. in 1979; pub. as 2n rev. ed. in 1985.Vol. 2C, Pg. 4628, 1982. | |
| rabbit | LD50 | skin | 5660uL/kg (5.66mL/kg) | Food and Cosmetics Toxicology. Vol. 11, Pg. 95, 1973. Link to PubMed | |
| rat | LD50 | intraperitoneal | 800mg/kg (800mg/kg) | "Patty's Industrial Hygiene and Toxicology," 3rd rev. ed., Clayton, G.D., and F.E. Clayton, eds., New York, John Wiley & Sons, Inc., 1978-82. Vol. 3 originally pub. in 1979; pub. as 2n rev. ed. in 1985.Vol. 2C, Pg. 4628, 1982. | |
| rat | LD50 | oral | 3560mg/kg (3560mg/kg) | Food and Cosmetics Toxicology. Vol. 11, Pg. 95, 1973. Link to PubMed |