Products Categories
| CAS No.: | 643-22-1 |
|---|---|
| Name: | Erythromycin stearate |
| Molecular Structure: | |
|
|
|
| Formula: | C37H67NO13.C18H36O2 |
| Molecular Weight: | 1018.40 |
| Synonyms: | Octadecanoic acid, compd. with erythromycin (1:1);Abboticine;Erythromycin,octadecanoate (salt);Bristamycin (TN);Erypar;Bristamycin;Qidmycin;[(2S,3R,4S,6R)-2-[[(3R,4S,5S,6R,7R,9R,11R,12R,13R,14R)-14-ethyl-7,12,13-trihydroxy-4-[(2S,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyl-oxan-2-yl]oxy-3,5,7,9,11,13-hexamethyl-2,10-dioxo-1-oxacyclotetradec-6-yl]oxy]-3-hydroxy-6-methyl-oxan-4-yl]-dimethyl-azanium; octadecanoate; |
| EINECS: | 211-396-1 |
| Density: | 1.112g/cm3 |
| Melting Point: | 77-79 °C |
| Boiling Point: | 986.7 °C at 760 mmHg |
| Flash Point: | 550.5 °C |
| Appearance: | fine white powder |
| Hazard Symbols: |
 Xn; Xn;  Xi Xi
|
| Risk Codes: | 42/43-36/37/38 |
| Safety: | 26-36 |
| PSA: | 231.21000 |
| LogP: | 8.11810 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 717878-06-31-(4-fluorophenyl)-4-nitro-1H-imidazole
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- 146447-26-9N-{4-[4-(Piperidinomethyl)pyridyl-2-oxy]-cis-2-butene}phthalimide maleic acid
- 106-49-0p-Toluidine
- 103129-82-4(S)-Amlodipine
- 96-23-11,3-Dichloro-2-propanol
- 69-89-6Xanthine
- 89-61-22,5-Dichloronitrobenzene
- 109-92-2Ethyl vinyl ether
- 61-82-5Triazol-3-amine
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin

What can I do for you?
Get Best Price
Consensus Reports
NTP Carcinogenesis Studies (Feed); No Evidence; rat, mouse NCITR* National Cancer Institute Carcinogenesis Technical Report Series. (Bethesda, MD 20014) No. NTP-TR-338 ,1988.
Specification
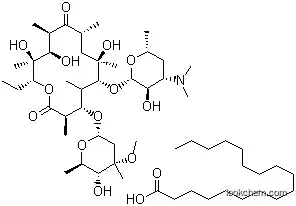
The Erythromycin stearate with CAS registry number of 643-22-1 is also called Abboticine. The IUPAC name is (3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-6-[(2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyloxan-2-yl]oxy-14-ethyl-7,12,13-trihydroxy-4-[(2R,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyloxan-2-yl]oxy-3,5,7,9,11,13-hexamethyl-oxacyclotetradecane-2,10-dione; octadecanoic acid. Its EINECS registry number is 211-396-1. In addition, the molecular formula is C37H67NO13.C18H36O2 and the molecular weight is 1018.40. It is a kind of white solid and belongs to the classes of Intermediates; Fine Chemicals; Pharmaceuticals; Antibacterial; Chemical Structure Class; Interferes with Protein SynthesisAntibiotics; Macrolides. What's more, it is a bacteriostatic antibiotic macrolide produced by Streptomyces erythreus.
Physical properties about Erythromycin stearate are: (1)ACD/LogP: 10.51; (2)# of Rule of 5 Violations: 4; (3)ACD/LogD (pH 5.5): 8.02; (4)ACD/LogD (pH 7.4): 8; (5)#H bond acceptors: 16; (6)#H bond donors: 6; (7)#Freely Rotating Bonds: 27; (8)Polar Surface Area: 231.21 Å2; (9)Flash Point: 550.5 °C; (10)Enthalpy of Vaporization: 163.03 kJ/mol; (11)Boiling Point: 986.7 °C at 760 mmHg; (12)Vapour Pressure: 0 mmHg at 25°C.
When you are using this chemical, please be cautious about it as the following:
This chemical may cause sensitization by inhalation and skin contact. And it is irritating to eyes, respiratory system and skin. During using it, wear suitable protective clothing. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: CCCCCCCCCCCCCCCCC(O)=O.CN(C)[C@H]3C[C@@H](C)O[C@@H](O[C@@H]2[C@@H](C)[C@H](O[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1)[C@@H](C)C(=O)O[C@H](CC)[C@@](C)(O)[C@H](O)[C@@H](C)C(=O)[C@H](C)C[C@@]2(C)O)[C@@H]3O
(2)InChI: InChI=1/C37H67NO13.C17H34O2/c1-14-25-37(10,45)30(41)20(4)27(39)18(2)16-35(8,44)32(51-34-28(40)24(38(11)12)15-19(3)47-34)21(5)29(22(6)33(43)49-25)50-26-17-36(9,46-13)31(42)23(7)48-26;1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17(18)19/h18-26,28-32,34,40-42,44-45H,14-17H2,1-13H3;2-16H2,1H3,(H,18,19)/t18-,19-,20+,21+,22-,23+,24+,25-,26+,28-,29+,30-,31+,32-,34+,35-,36-,37-;/m1./s1
(3)InChIKey: SUDWFYYCHWBVFZ-YZPBMOCRBF
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | intraperitoneal | 413mg/kg (413mg/kg) | Drugs in Japan Vol. -, Pg. 211, 1990. | |
| mouse | LD50 | oral | 3112mg/kg (3112mg/kg) | Drugs in Japan Vol. -, Pg. 211, 1990. | |
| mouse | LD50 | subcutaneous | > 2500mg/kg (2500mg/kg) | Drugs in Japan Vol. -, Pg. 211, 1990. | |
| women | TDLo | oral | 60mg/kg (60mg/kg) | GASTROINTESTINAL: CHANGES IN STRUCTURE OR FUNCTION OF ENDOCRINE PANCREAS GASTROINTESTINAL: NAUSEA OR VOMITING | Journal of Toxicology, Clinical Toxicology. Vol. 34, Pg. 93, 1996. |