Y. Uozumi et al.
[
4
(PdCl
4.17, H 4.03, N 6.72; found: C 44.11,
H 4.15, N 7.18.
Preparation of a PdCl
2
)
3
A
H
U
G
R
N
U
G
7
H
7
N)
6
·CH
3
CO
2
Et]
n
:
C
2
/polyviologen
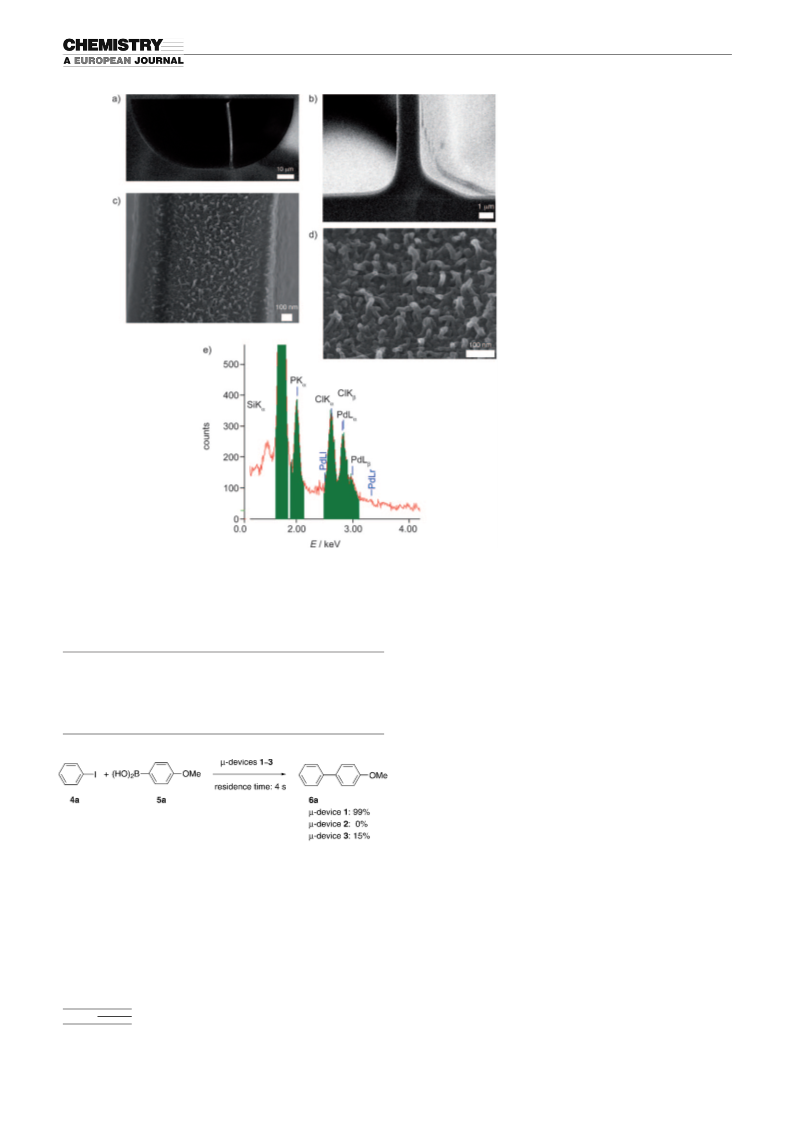
membrane inside a microchannel (m-
device 3): A solution of poly{(4,4’-bi-
pyridyl)-co-[1,4-bis(bromomethyl)ben-
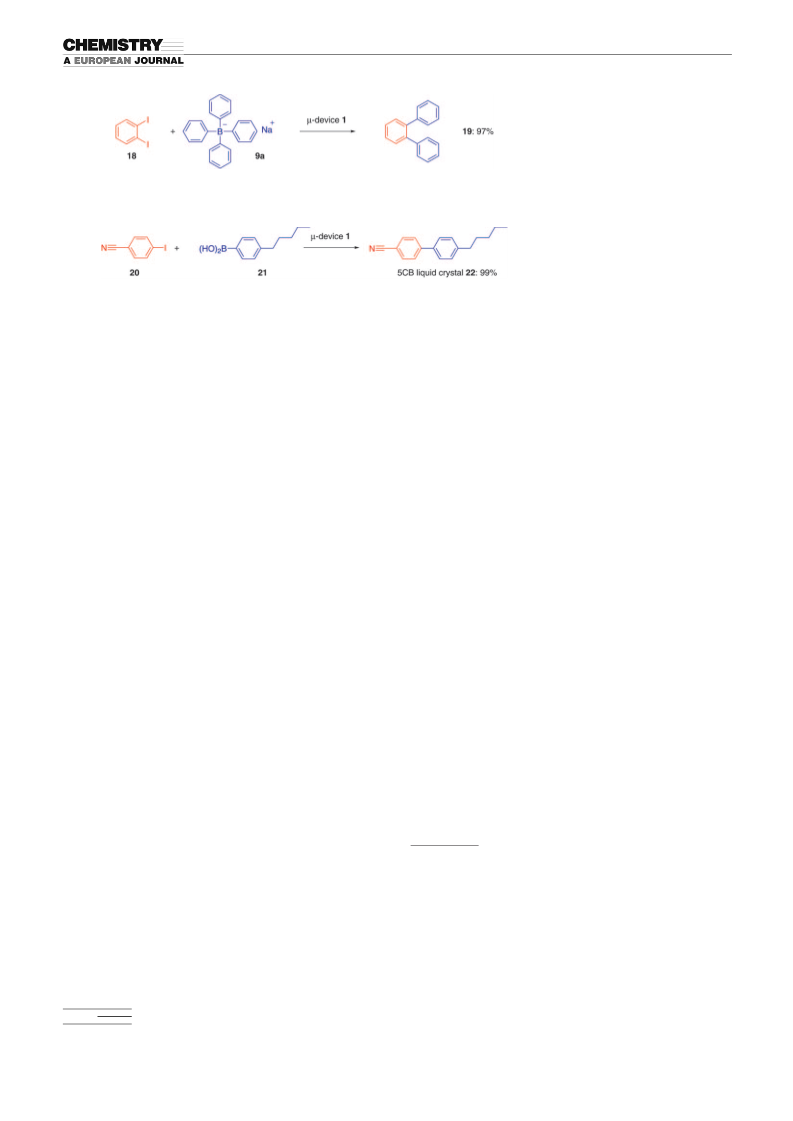
Scheme 8. Preparation of o-terphenyl.
zene]} in iPrOH/EtOAc/H
3.4 mm pyridinium unit; solution A)
and an aqueous solution of PdCl
1.7 mm) and NaCl (17 mm) (solu-
2
O (2:1:1)
(
2
(
tion B) were charged oppositely into
the microchannel at 258C for 5 min
À1
with a flow rate of 25 mLmin . Two-
phase parallel laminar flow was
formed under the flowing conditions,
Scheme 9. Preparation of a 5CB liquid crystal.
and brownish polymer membrane 3
was precipitated out at the interface
between the two parallel flows (thickness of membrane 3=10 mm).
was readily obtained quantitatively from the outlet of m-
device 1. (Schemes 6–9)
Representative procedure for cross-coupling using the microchannel with
a divided PA–TAP–Pd membrane (Table 2, entry 6; m-device 1): A solu-
tion of 4-trifluoromethyliodobenzene (4c) in EtOAc/iPrOH (2:5; 6.3 mm;
solution C)) and a solution of 4-methoxyphenylboronic acid (5a) and
Na CO in water (ArB(OH) =9.4 mm, Na CO =18.3 mm; solution D)
Conclusion
2
3
2
2
3
were oppositely introduced into the membrane-divided channel m-
À1
device 1 at 508C with a flow rate of 2.5 mLmin (for solution C) and
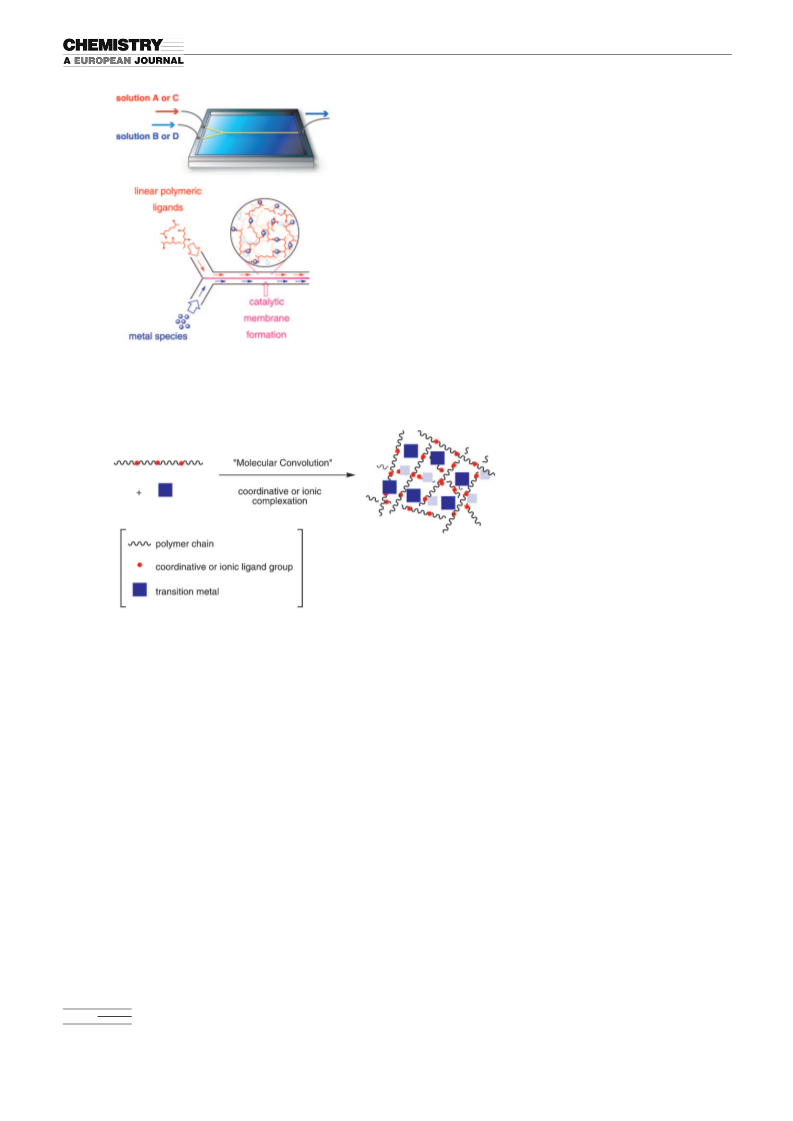
A variety of palladium membranes were installed inside mi-
crochannel reactors through the “ship-in-a-bottle” protocol
of our molecular convolution method to provide microreac-
tion devices. These microchannel devices were applied to
the instantaneous Suzuki–Miyaura reaction and allyl–aryl
coupling reaction to afford the corresponding coupling prod-
ucts in quantitative yields within 5 and 1 s of residence time,
respectively. m-Device 1 was applied to the preparation of
bioactive compounds as well as functional materials to give
the target molecules in high yield. The extension of catalytic
membrane-installed microchannel reactors with a variety of
convoluted polymeric metal complexes to other organic
transformations is currently in progress.
À1
5
.0 mLmin (for solution D), respectively, and two parallel laminar
layers flowed through the channel in 4 s. The resulting organic/aqueous
microstream was collected from the outlet of the channel to afford a
À1
quantitative yield (240 mgh ) of 4-methoxy-4’-(trifluoromethyl)biphenyl
(
6 f). The chemical yield and structure of the product 6 f were determined
by GC (based on similarity of GC–MS data with the National Institute of
1
Standards and Technology (NIST) mass database library) and H NMR
spectroscopy.
Representative procedure for the allylic arylation reaction using the mi-
crochannel with a divided PA–TAP–Pd membrane (Table 5, entry 1; m-
device 1): A solution of cinnamyl acetate (12a) in iPrOH (5.8 mm; solu-
tion C) and an aqueous solution of sodium tetraphenylborate (9a)
(
58 mm; solution D) were oppositely introduced into the membrane-di-
À1
vided channel m-device 1 at 708C with a flow rate of 3.0 mLmin , and
two parallel laminar layers flowed through the channel in 1 s. The result-
ing organic/aqueous microstream was collected from the outlet of the
channel to afford a quantitative yield of 1,3-diphenyl-1-propene (13a).
Experimental Section
Acknowledgements
Preparation of a PdCl
device 1): An solution of PA-TAP in EtOAc (5.0 mm phosphorus unit;
solution A) and an aqueous solution of [PdCl (NH ] (1.7 mm; solu-
tion B) were charged oppositely into the microchannel (100 mm width,
2
/PA-TAP membrane inside a microchannel (m-
This work was supported by the Green-Sustainable Chemical Process
project sponsored by the METI/NEDO. We also thank the JSPS (Grant-
in-Aid for Scientific Research no. 20655035), the MEXT (Science Re-
search on Priority Areas no. 460), the JST (CREST Project), and the
NEDO (GSC Project) for partial financial support of this work. We are
thankful to Dr. Satoru Nakao (IMS) for his help in measuring the SEM,
which is supported by the Nanotechnology Support Project (MEXT) and
managed by the IMS.
4
A
T
N
T
E
N
N
4 2
)
4
2
0 mm depth, 40–140 mm length) at 258C for 10 min with a flow rate of
5 mLmin . Two-phase parallel laminar flow was formed under the flow-
À1
ing conditions and yellowish polymer membrane 1 was precipitated out
at the interface between the two parallel flows (thickness of membrane
1
=1 mm).
Elemental analysis
calcd
(%)
for
(
C
100
H
144
O
10
N
10
P
2
PdCl ·5H O) : C 60.79, H 7.86, N 7.09, P 3.14, Pd 5.39;
2
2 n
found: C 60.36, H 7.67, N 6.97, P 2.99, Pd 5.79 (Pd and P were measured
with ICP-AES).
[
1] For a recent review of solid-phase reactions by using palladium cata-
lysts, see: a) Y. Uozumi, T. Hayashi in Handbook of Combinatorial
Chemistry (Eds.: K. C. Nicolaou, R. Hanko, W. Hartwig) Wiley-
VCH, Weinheim, 2002, Chapter 19; b) T. Franzel, W. Solodenko, A.
Kirschning in Polymeric Materials in Organic Synthesis and Catalysis
(Ed.: M. R. Buchmeiser), Wiley-VCH, Weinheim, 2003, Chapter 4;
Preparation of a PdCl
device 2): An solution of poly(4-vinylpyridine) in EtOAc (5.0 mm pyri-
dine unit; solution A) and an aqueous solution of PdCl (1.7 mm) and
2
/polyPy membrane inside a microchannel (m-
2
NaCl (17 mm) (solution B) were charged oppositely into the microchan-
À1
nel at 258C for 5 min with a flow rate of 25 mLmin . Two-phase parallel
laminar flow was formed under the flowing conditions and yellowish
polymer membrane 2 was precipitated out at the interface between the
two parallel flows (thickness of membrane 2=10 mm). IR (ATR) n˜ =
À1
3
476, 1612, 1427, 1221, 1068, 829 cm ; elemental analysis calcd (%) for
11318
ꢀ 2010 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Chem. Eur. J. 2010, 16, 11311 – 11319

 Yamada, Yoichi M. A.
Yamada, Yoichi M. A.
 Watanabe, Toshihiro
Watanabe, Toshihiro
 Beppu, Tomohiko
Beppu, Tomohiko
 Fukuyama, Naoshi
Fukuyama, Naoshi
 Torii, Kaoru
Torii, Kaoru
 Uozumi, Yasuhiro
Uozumi, Yasuhiro