Organometallics
Article
31P{1H} NMR (CDCl3): δ −16.9 (s). IR (Nujol, cm−1): νmax 3055 br
m, 2605 w, 2125 w, 1570 w, 1540 s, 1499 w, 1314 w, 1295 w, 1238 m,
1164 s, 1089 s, 1074 s, 1058 s, 1049 s, 1025 s, 956 w, 912 w, 890 m,
879 m, 833 w, 815 m, 790 s, 754 s, 733 s, 702 s, 635 m, 587 m, 501 s,
447 m. ESI+ MS: m/z 383 ([Ph2PfcCH2]+); ESI− MS: m/z 165
([C6H4(CO2H)CO2]−), 546 ([3 − H2O − H]−). Anal. Calcd for
C31H28FeNO4P (565.4): C, 65.86; H, 4.99; N, 2.48. Found: C, 65.55;
H, 4.89; N, 2.30.
NEt3]+), 924 ([7 − Cl]+); ESI− MS: m/z 582 ([2 − HNEt3]−), 857
([7 − HNEt3])−). Anal. Calcd for C45H53N3ClFeO4PPdS (960.7): C,
56.26; H, 5.56; N, 4.37. Found: C, 55.98; H, 5.64; N, 4.19.
Synthesis of Complex 8. This complex was obtained similarly,
starting from 2 (68.4 mg, 0.100 mmol) and [(C6H4C6H4NH2)PdCl]2
(31.0 mg, 0.050 mmol), and was isolated as an orange solid in
quantitative yield.
1H NMR (CDCl3): δ 1.23 (t, 3JHH = 7.3 Hz, 9 H, CH3 of HNEt3),
3.05 (q, 3JHH = 7.3 Hz, 6 H, CH2 of HNEt3), 3.28 (br s, 1 H, fc), 3.89
(br s, 1 H, fc), 3.91 (br s, 1 H, fc), 4.08 (br s, 1 H, fc), 4.14−4.28 (m, 4
H, 2 H of fc and 2 H of CH2NHCO), 4.50 (br s, 1 H, fc), 4.95 (br s, 1
H, NH2), 5.01 (br s, 1 H, fc), 5.24 (br s, 1 H, NH2), 6.45 (t, J = 7.3 Hz,
1 H, C6H4Pd), 6.58 (t, J = 6.8 Hz, 1 H, C6H4Pd), 6.91 (t, J = 7.3 Hz, 1
H, C6H4Pd), 7.01 (m, 2 H, C6H4NH2), 7.10 (m, 2 H, C6H4NH2),
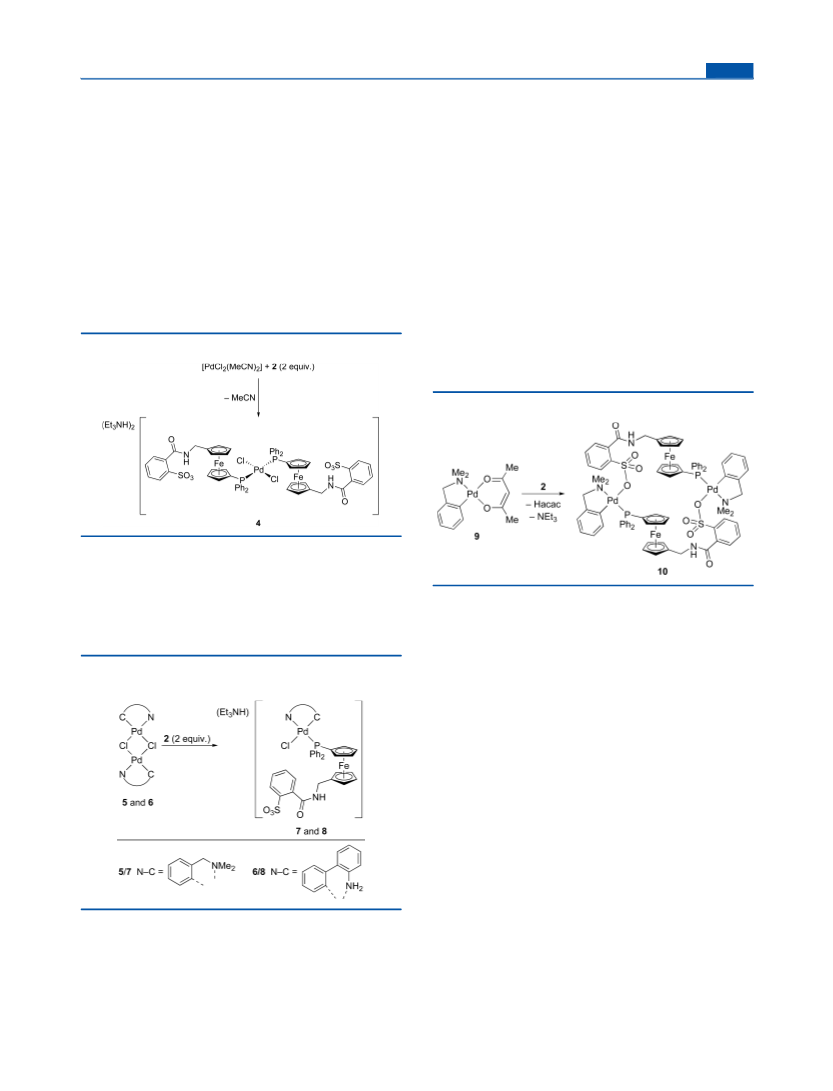
Synthesis of [PdCl2(2-κP)2] (4). Ligand 2 (41.1 mg, 0.060 mmol)
and [PdCl2(MeCN)2] (7.8 mg, 0.030 mmol) were dissolved in dry
dichloromethane (5 mL). After it was stirred for 60 min, the deep red
reaction mixture was filtered through a PTFE syringe filter (0.45 μm
pore size) and evaporated under vacuum to afford 4 as a gummy red
solid in essentially quantitative yield. The compound tends to retain
the reaction solvent.
7.18−7.26 (m, 3 H, 1 H of C6H4SO3 and 2 H of PPh2), 7.29 (d, JHH
=
3
1H NMR (CDCl3): δ 1.19 (t, JHH = 7.3 Hz, 9 H, CH3 of HNEt3),
7.4 Hz, 1 H, C6H4Pd), 7.38−7.43 (m, 5 H, 1 H of C6H4SO3 and 4 H
of PPh2), 7.62 (dd, J = 7.1, 1.8 Hz, 1 H, C6H4SO3), 7.62−7.74 (m, 4
H, PPh2), 7.96 (br s, 1 H, NHCO), 8.03 (m, 1 H, C6H4SO3). The
signal due to HNEt3 is extremely broadened. 13C{1H} NMR (CDCl3):
δ 8.59 (CH3 of HNEt3), 39.37 (CH2NHCO), 46.20 (CH2 of HNEt3),
2.99 (q, 3JHH = 7.3 Hz, 6 H, CH2 of HNEt3), 4.32 (d, 3JHH = 5.4 Hz, 2
H, CH2NHCO), 4.49 (vt, J′ = 1.9 Hz, 2 H, fc), 4.50 (vt, J′ = 1.9 Hz, 2
H, fc), 4.55 (m, 4 H, fc), 7.34−7.44 (m, 6 H, PPh2), 7.59−7.65 (m, 4
3
H, PPh2), 7.71 (m, 2 H, C6H4), 8.02 (m, 2 H, C6H4), 8.31 (t, JHH
=
68.79 (CH of fc), 70.22 (CH of fc), 70.31 (CH of fc), 71.58 (d, 1JPC
=
=
5.4 Hz, 1 H, NHCO). Signal due to HNEt3 was not observed due to
broadening. 13C{1H} NMR (CDCl3): δ 8.68 (CH3 of HNEt3), 39.42
(CH2NHCO), 46.24 (CH2 of HNEt3), 69.69 (CH of fc), 71.10 (CH
of fc), 71.41 (apparent t, J = 28 Hz, Cipso-P of fc), 72.95 (apparent t, J
= 3 Hz, CH of fc), 75.86 (apparent t, JPC = 6 Hz, CH of fc), 86.56
(Cipso-CH2 of fc), 127.08 (CH of C6H4), 127.69 (apparent t, JPC = 5
Hz, CH of PPh2), 129.50 (CH of C6H4), 130.17 (CHpara of PPh2),
130.29 (CH of C6H4), 131.44 (apparent t, J = 25 Hz, Cipso of PPh2),
134.14 (apparent t, JPC = 6 Hz, CH of PPh2), 134.54 (CH of C6H4),
142.04 (Cipso of C6H4), 168.58 (CO). The signal due to one Cipso of
C6H4 is probably obscured by other, more intense signals. 31P{1H}
NMR (CDCl3): δ +16.3 (s). IR (DRIFTS, cm−1): νmax 3444 br m,
3297 br m, 3055 m, 2984 m, 1648 s, 1552 m, 1481 m, 1436 s, 1303 m,
1235 s, 1193 s, 1098 w, 1082 w, 1019 s, 837 w, 748 m, 696 s, 617 s,
568 m, 541 m, 506 s, 475 m. ESI+ MS: m/z 1271 ([Pd-
(Ph2PfcCH2NHCOC6H4SO3)2 + H]+); ESI− MS: m/z 1343 ([4 +
H − 2HNEt3]+). Anal. Calcd for C72H82N4Cl2Fe2O8PdP2S2·1.5CH2Cl2
(1673.8): C, 52.74; H, 5.12; N, 3.35. Found: C, 52.99; H, 5.33; N,
3.27.
5 Hz, Cipso-P of fc), 72.44 (CH of fc), 73.03 (CH of fc), 73.42 (d, JPC
8 Hz, CH of fc), 73.76 (d, JPC = 7 Hz, CH of fc), 75.84 (d, JPC = 13
Hz, CH of fc), 86.17 (Cipso-CH2 of fc), 120.50 (CH of C6H4NH2),
124.84 (CH of C6H4Pd), 125.67 (CH of C6H4Pd), 125.96 (CH of
C6H4SO3), 127.07 (d, 3JPC = 5 Hz, CH of C6H4Pd), 127.18 (CHpara of
PPh2), 127.38 (d, JPC = 11 Hz, CH of C6H4NH2), 128.15 (CH of
C6H4SO3), 129.45 (CH of C6H4SO3), 129.74 (CH of C6H4SO3),
129.86 (d, 2JPC = 18 Hz, CHortho of PPh2), 130.70 (CH of C6H4NH2),
130.71 (d, 1JPC = 52 Hz, Cipso of PPh2), 132.21 (d, 2JPC = 47 Hz, Cipso
3
of C6H4Pd), 133.41 (d, JPC = 10 Hz, Cipso of C6H4NH2), 134.57 (d,
3JPC = 13 Hz, CHmeta of PPh2), 135.41 (Cipso of C6H4SO3), 138.04
(CH of C6H4NH2), 139.71 (d, 3JPC = 10 Hz, CH of C6H4Pd), 139.94
(Cipso of C6H4NH2), 141.91 (Cipso of C6H4SO3), 151.53 (Cipso of
C6H4Pd), 168.70 (CO). 31P{1H} NMR (CDCl3): δ +30.3 (s). IR
(Nujol, cm−1): νmax 3435 br w, 3306 br w, 3050 w, 1644 s, 1591 m,
1558 s, 1302 m, 1228 s, 1181 s, 1168 s, 1138 m, 1098 m, 1081 m, 1059
w, 1017 s, 836 w, 792 w, 748 s, 694 s, 658 w, 615 s, 567 m, 523 m, 501
m, 470 m. ESI+ MS: m/z 857 ([8 − Cl − NEt3]+), 879 ([8 + Na −
HNEt3 − Cl]+), 895 ([8 + K − HNEt3 − Cl]+), 958 ([8 − Cl]+);
ESI− MS: m/z 582 ([2 − HNEt3]−), 688 ([Pd(2 − HNEt3)]−). Anal.
Calcd for C48H51N3ClFeO4PPdS·0.5CH2Cl2 (1037.1): C, 56.16; H,
5.05; N, 4.05. Found: C, 56.25; H, 5.19; N, 4.04.
Synthesis of [(LNC)PdCl(2-κP)] (7). Ligand 2 (68.4 mg, 0.10
mmol) and [(LNC)PdCl]2 (27.6 mg, 0.050 mmol) were dissolved in
dry dichloromethane (5 mL). The resulting solution was stirred for 60
min and then evaporated under vacuum to afford complex 5 as a
yellow-orange foam in virtually quantitative yield.
Synthesis of Complex 10. Compound 2 (68.4 mg, 0.10 mmol)
and [(LNC)Pd(acac)] (33.9 mg, 0.10 mmol) were mixed in
dichloromethane (5 mL), and the reaction mixture was stirred for
60 min, during which time all of the solid educts dissolved. The orange
solution was filtered through a silica gel column, with dichloro-
methane−methanol (10:1) as eluent. Subsequent evaporation
furnished 10 as a yellow solid (73.5 mg, 89%). The orange crystals
used for the structure determination were obtained from methanol−
diethyl ether.
3
1H NMR (CDCl3): δ 1.23 (t, JHH = 7.3 Hz, 9 H, CH3 of HNEt3),
2.83 (d, 4JPH = 2.0 Hz, 6 H, NMe2), 3.04 (q, 3JHH = 7.3 Hz, 6 H, CH2
4
of HNEt3), 4.12 (d, JPH = 2.0 Hz, 2 H, CH2NMe2), 4.35 (d, J′ = 2.0
Hz, 2 H, fc), 4.43 (vtd, J′ = 1.9 Hz, 1.0 Hz, 2 H, fc), 4.54 (s, 4 H, fc),
6.27 (m, 1 H, C6H4Pd), 6.39 (m, 1 H, C6H4Pd), 6.81 (td, J = 7.3 Hz,
1.1 Hz, 1 H, C6H4Pd), 7.00 (dd, J = 7.4 Hz, 1.5 Hz, 1 H, C6H4Pd),
7.28−7.34 (m, 3 H, PPh2), 7.37−7.43 (m, 5 H, 2 H of C6H4SO3 and 3
H of PPh2), 7.52−7.58 (m, 4 H, PPh2), 7.67 (m, 1 H, C6H4SO3), 8.03
(m, 1 H, C6H4SO3), 8.15 (br s, 1 H, NHCO), 9.66 (br s, HNEt3).
13C{1H} NMR (CDCl3): δ 8.58 (CH3 of HNEt3), 39.44
1H NMR (CDCl3): δ 2.56 (br s, 6 H, NMe2), 3.98 (br s, 2 H) and
4.25−4.57 (br m, 10 H, CH2 and fc), 6.24 (t, J = 7.2 Hz, 1 H,
C6H4Pd), 6.32 (br s, 1 H, C6H4Pd), 6.78 (br s, 1 H, C6H4Pd), 6.89 (br
s, 1 H, C6H4Pd), 7.28−7.50 (br m, 8 H, PPh2 and/or C6H4SO3), 7.68
(br s, 4 H, PPh2 and/or C6H4SO3), 7.86 (br s, 1 H, C6H4SO3), 8.08
(br s, 1 H, C6H4SO3), 9.15 (br s, 1 H, CONH). 31P{1H} NMR
(CDCl3): δ +33.8 (br s). 13C NMR spectra could not be recorded for
solubility reasons. IR (Nujol, cm−1): νmax 3435 m, 3327 br m, 3052 m,
1649 s, 1580 m, 1564 m, 1546 m, 1301 m, 1248 m, 1182 w, 1162 s,
1135 w, 1099 m, 1076 m, 1019 m, 1004 m, 990 m, 845 m, 812 w, 790
w, 744 s, 697 s, 657 w, 617 s, 564 m, 545 m, 502 m, 487 m, 446 w.
E S I − M S : m / z 8 2 3 ( [ ( C 6 H 4 C H 2 N M e 2 ) P d -
(Ph2PfcCH2NHCOC6H4SO3 + H]+), 845 and 861 (the same as
Na/K+); ESI− MS: m/z 582 ([2 − HNEt3]−). Anal. Calcd for
C39H37N2FeO4PPdS (823.0): C, 56.91; H, 4.53; N, 3.40. Found: C,
56.70; H, 4.77; N, 3.58.
3
(CH2NHCO), 46.23 (CH2 of HNEt3), 50.16 (d, JPC = 2 Hz,
NMe2), 69.72 (CH of fc), 71.20 (CH of fc), 72.80 (d, JPC = 7 Hz, CH
of fc), 72.94 (Cipso-P of fc, partially overlapped), 73.54 (CH2NMe2),
75.75 (d, JPC = 10 Hz, CH of fc), 86.36 (Cipso-CH2 of fc), 122.49 (CH
of C6H4Pd), 123.69 (CH of C6H4Pd), 124.78 (d, JPC = 6 Hz,
C6H4Pd), 127.10 (CH of C6H4SO3), 127.82 (d, JPC = 11 Hz, CH of
PPh2), 129.48 (CH of C6H4SO3), 130.12 (Cipso of C6H4SO3), 130.17
(CH of C6H4SO3), 130.41 (d, 4JPC = 2 Hz, CHpara of PPh2), 131.83 (d,
1JPC = 50 Hz, Cipso of PPh2), 134.33 (d, JPC = 12 Hz, CH of PPh2),
134.58 (Cipso of C6H4SO3), 138.37 (d, 3JPC = 11 Hz, CH of C6H4Pd),
2
141.93 (CH of C6H4SO3), 148.22 (d, JPC = 2 Hz, Cipso of C6H4Pd),
152.04 (br s, Cipso of C6H4Pd), 168.63 (CO). 31P{1H} NMR
(CDCl3): δ +33.1 (s). IR (Nujol, cm−1): νmax 3462 br m, 3287 br m,
3047 m, 1648 s, 1560 m, 1303 m, 1234 m, 1189 m, 1165 m, 1139 w,
1099 w, 1082 w, 1019 m, 845 m, 745 m, 696 m, 658 w, 616 m, 567 w,
544 w, 524 w, 504 m, 477 m, 439 w. ESI+ MS: m/z 823 ([7 − Cl −
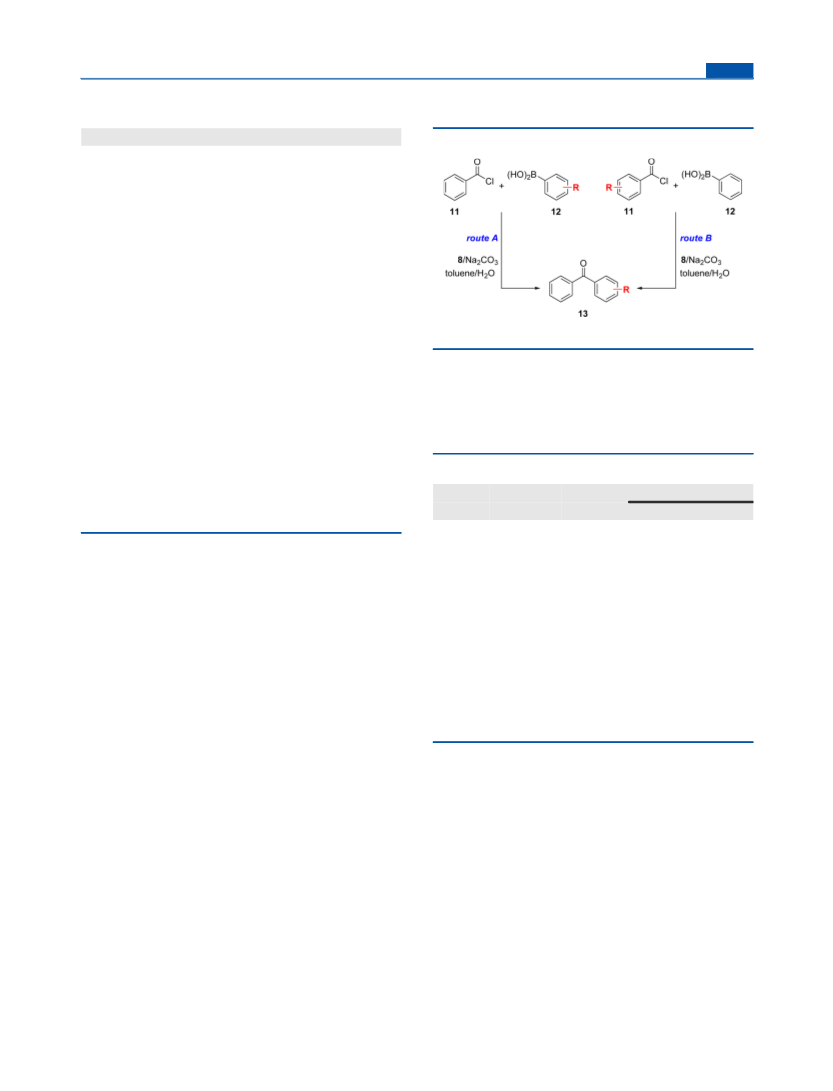
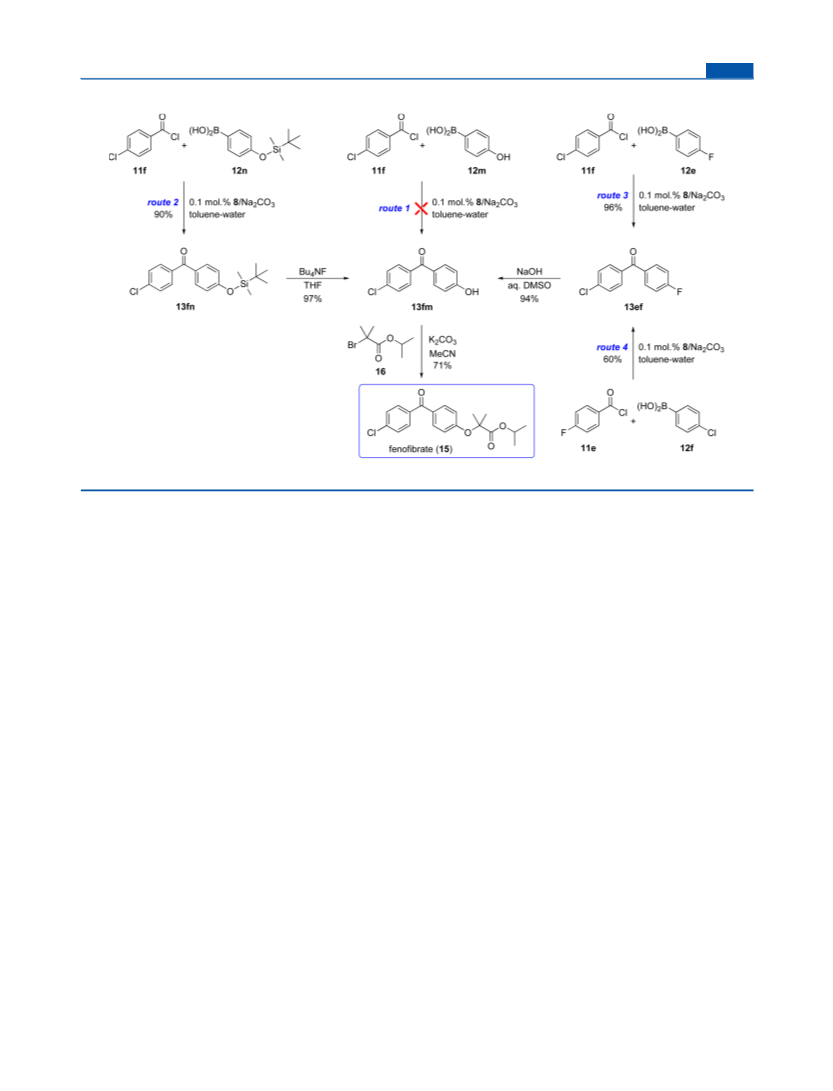
Catalytic Experiments: General Procedure. A Schlenk tube was
successively charged with a stirring bar, the respective acyl chloride
H
Organometallics XXXX, XXX, XXX−XXX

 ?koch, Karel
?koch, Karel
 Císa?ová, Ivana
Císa?ová, Ivana
 ?těpni?ka, Petr
?těpni?ka, Petr