582
D. Haddenham et al. / Tetrahedron 67 (2011) 576e583
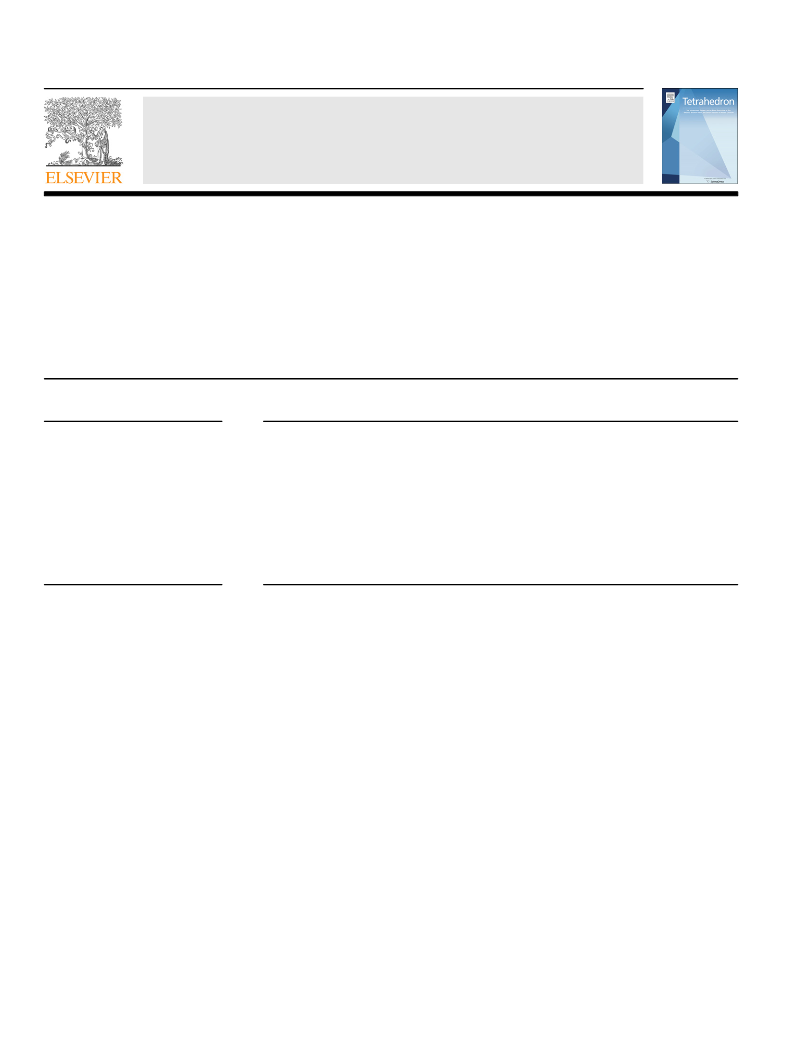
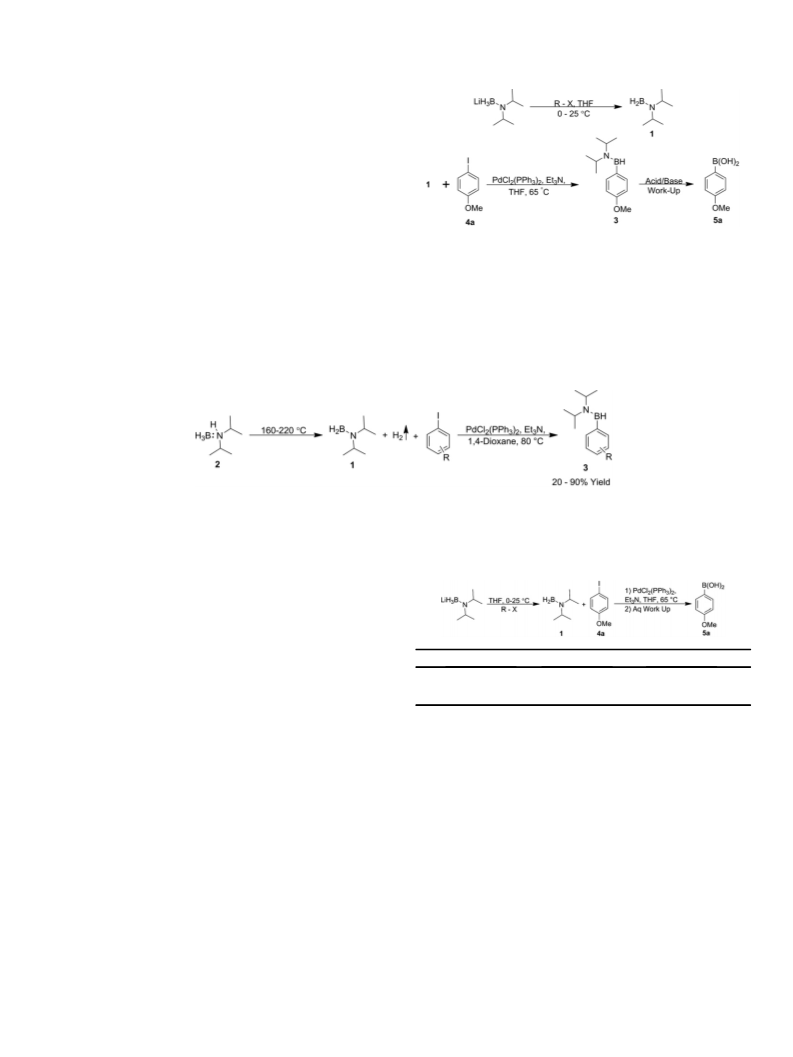
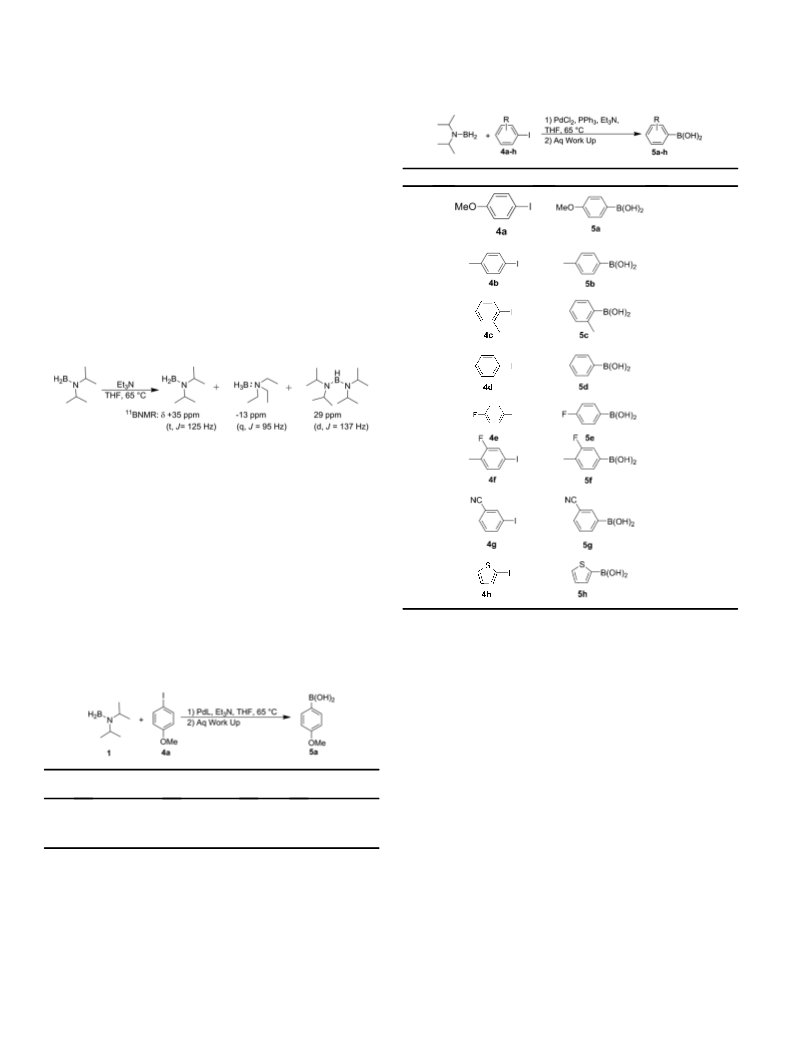
1
43.1; 11B NMR (160 MHz, MeOH)
for C 165.11054, found 165.10814.
d
þ29.1; EM (ESI): m/z (Mþ) calcd
MeOD)
found 176.08774.
d
þ28.3; EM (ESI): m/z (Mþ) calcd for C
9
H11BNO
2
176.09440,
9 2
H14BO
21
1
21
1
4
.4.3. 2-Tolylboronic acid (5c) (7c) (9c) . Off-white solid, H NMR
4.4.14. Naphthalen-2-ylboronic acid (9i) . Off-white solid, H NMR
(
500 MHz, CDCl
3
)
d
2.86 (s, 3H), 7.31 (d, 1H, J¼8.0 Hz), 7.35 (t, 1H,
(500 MHz, MeOH-d
4
)
d
7.48 (m, 2H), 7.76e7.89 (m, 4H), 8.24 (m,
1H); C NMR (125 MHz, MeOH-d 126.8,127.7,128.7,129.6,131.4,
þ29.3; EM (ESI): m/z (Mþ)
223.09631, found 223.09008.
13
13
J¼7.5 Hz), 7.49 (t, 1H, J¼7.5 Hz), 8.26 (d, 1H, J¼7.0 Hz); C NMR
4
) d
4
) d
11
11
(
(
C
125 MHz, CDCl
160 MHz, MeOH)
13BNaO 187.10383, found 187.09008.
3
)
d
23.3, 125.4, 130.8, 132.4, 137.5, 146.5; B NMR
135.7; B NMR (160 MHz, MeOH-d
calcd for C12 13BNaO
d
þ28.5; EM (ESI): m/z (Mþ) calcd for
H
2
9
H
2
4
.4.15. 4,4,5,5-tetramethyl-2-p-tolyl-1,3,2-dioxaborolane
2
1
1
23
1
4
.4.4. Phenylboronic acid (5d) (7d) (9e) . Off-white solid, H NMR
500 MHz, CDCl
7.54 (t, 2H, J¼7.5 Hz), 7.62 (t, 1H, J¼7.5 Hz), 8.27
d, 2H, J¼7.0 Hz); C NMR (125 MHz, CDCl
1
3
(10a) . Yellow Oil, H NMR (500 MHz, CDCl ) d 1.38 (s, 8H), 2.40 (s,
13
(
(
3
)
d
3H), 7.23 (d, 2H, J¼8.0 Hz), 7.77 (d, 2H, J¼8 Hz); C NMR (125 MHz,
1
3
11
3
)
d
128.1, 132.9, 135.8;
þ28.5; EM (ESI): m/z (Mþ) calcd for
151.08092, found 151.09249.
CDCl
CDCl
3
)
)
d
21.8, 24.9, 83.7, 128.7, 135.0, 141.5; B NMR (160 MHz,
þ31.3.
1
B NMR (160 MHz, MeOH)
d
3
d
C
8
H12BO
2
4
.4.16. 2-(4-methylphenyl)-5,5-dimethyl-1,3,2-dioxaborinane
21
6b,7a,20a
1
4
.4.5. 4-Fluorophenylboronic acid (5e) (7e) (9g) . Off-white solid,
(10b)
3
. Yellow Oil, H NMR (500 MHz, CDCl ) d 1.06 (s, 6H),
1
H NMR (500 MHz, CDCl
3
)
d
7.21 (t, 2H, J¼9.0 Hz), 8.24 (t, 2H,
2.41 (s, 3H), 3.80 (s, 4H), 7.23 (d, J¼8.0 Hz), 7.77 (d, 2H, J¼6.0 Hz);
13
13
J¼8.5 Hz); C NMR (125 MHz, MeOD) 112.2 (d, J ¼20 Hz),134.0 (d,
d
F
C NMR (125 MHz, CDCl
3
)
d
21.7, 22.0, 31.9, 72.4, 128.5, 134.1, 140.8;
þ27.1; EM (ESI): m/z (Mþ) calcd for
1
1
11
J
F
¼8 Hz) 162.7 (d, J
F
¼246 Hz); B NMR (160 MHz, MeOD)
d
þ28.5;
B NMR (160 MHz, CDCl
3
)
d
EM (ESI): m/z (Mþ) calcd for C
8
H
11BO
2
F 169.08972, found 169.08307.
12 2
C H18BO 205.14448, found 205.13944.
1
4
.4.6. 3-Fluoro-4-methylphenylboronic acid (5f). Off-white solid, H
4.4.17. 2-(4-methoxyphenyl)-5,5-dimethyl-1,3,2-dioxaborinane
6
b,7a,20a
1
NMR (500 MHz, MeOH-d
s, 1H), 7.35 (s, 1H); 13C NMR (125 MHz, MeOH-d
) d
4
4
)
d
2.21 (s, 3H), 7.13 (t, 1H, J¼7.0 Hz), 7.29
14.7, 120.7,
¼242); B NMR (160 MHz, MeOH-d
þ28.8; EM (ESI): m/z (Mþ) calcd for C 13BFO 183.10495, found
(12a)
3
. Yellow Oil, H NMR (500 MHz, CDCl ) d 1.02 (s, 6H),
(
3.76 (s, 4H), 3.80 (s, 3H), 6.92 (d, 2H, J¼8.5 Hz), 7.83 (d, 2H, J¼9 Hz);
11
13
120.8, 130.6, 132.1, 162.6 (d, J
F
4
)
C NMR (125 MHz, CDCl
3
)
d
22.0, 32.0, 55.1, 72.4,113.4,128.9, 132.4,
135.9,162.1; B NMR (160 MHz, CDCl
þ27.0; EM (ESI): m/z (Mþ)
calcd for C12 221.12795, found 221.13435.
11
d
9
H
2
3
) d
183.09872.
H18BO
3
4
.4.7. 3-Cyanophenylboronic acid (5g)22. Off-white solid, 1H NMR
7.45 (t, 1H, J¼8.0 Hz), 7.67 (d, 1H, J¼8.0 Hz)
.96 (m, 1H); 1 C NMR (125 MHz, MeOH-d
111.2, 118.7, 128.1,
þ27.6.
4.4.18. 2-(4-methylphenyl)-5,5-dimethyl-1,3,2-dioxaborinane
6
b,7a,20a
1
(
500 MHz, MeOH-d
4
)
d
(12b)
3
. Yellow Oil, H NMR (500 MHz, CDCl ) d 1.10 (s, 6H),
3
13
7
1
4
)
d
2.41 (s, 3H), 3.80 (s, 4H), 7.23 (d, J¼7.5 Hz), 7.78 (d, 2H, J¼5.0 Hz);
C
B
33.0, 137.1, 137.9; 11B NMR (160 MHz, MeOH-d
)
d
NMR (125 MHz, CDCl
NMR (160 MHz, CDCl
12 2
C H18BO 205.14448, found 205.13944.
11
4
3
) d 21.7, 22.0, 31.9, 72.4, 128.5, 134.1, 140.8;
3
)
d
þ27.1; EM (ESI): m/z (Mþ) calcd for
21
1
4.4.8. Thiopen-2-ylboronic acid (5h) (7g) . Off-white solid, H NMR
13
(
4
500 MHz, MeOH-d ) d 7.13 (s, 1H), 7.59 (s, 2H); C NMR (125 MHz,
11
MeOH-d
4
) 127.4, 130.9; B NMR (160 MHz, MeOH-d
4
)
d
þ26.8; EM
4.4.19. 5,5-dimethyl-2-(thiophen-2-yl)-1,3,2-dioxaborinane
6
b,7a,20a
1
(
ESI): m/z (Mþ) calcd for C
H
4 5
BO S 157.05865, found 157.04891.
2
(12c)
(
3
. Yellow Oil, H NMR (500 MHz, CDCl ) d1.04(s, 6H), 3.77
13
s, 4H), 7.20 (s,1H), 7.58 (s,1H), 7.63(s,1H); C NMR (125MHz, CDCl )
d 21.9, 72.5,128.2,131.5,135.8; B NMR (160MHz, CDCl ) d
3
(ESI): m/z (Mþ) calcd for C
3
.4.9. (E)-Styrylboronic acid (7f) . Off-white solid, 1H NMR
6.18 (d, 1H, J¼18.0 Hz), 7e7.3 (m, 5H) 7.42
126.6, 127.6,
2
1
11
þ25.5;EM
9 2
H14BO S 197.08817, found 197.08021.
4
(
500 MHz, MeOH-d
4
)
d
1
3
(
1
d, 1H, J¼7.0 Hz); C NMR (125 MHz, MeOH-d
4
) d
11
27.9, 128.3, 137.8, 146.9; B NMR (160 MHz, MeOH-d
4
)
d
þ32.4;
References and notes
EM (ESI): m/z (Mþ) calcd for C
H
8 9
BNaO
2
171.05905, found
171.05878.
1. (a) Miyaura, N.; Yanagi, T.; Suzuki, A. Synth. Commun. 1981, 11, 513; (b) Miyaura,
N.; Suzuki, A. Chem. Rev. 1995, 95, 2457; (c) Ishihara, K.; Yamamoto, H. Eur. J. Org.
Chem. 1999, 527; (d) Matteson, D. S. In Stereodirected Synthesis with Organo-
boranes; Hafner, K., Rees, C. W., Trost, B. M., Lehn, J. M., Von Rague Schleyer, P.,
Eds.; Springer: Heidelberg, 1995; (e) Bertounesque, E.; Florent, J. C.; Monneret,
C. Synthesis 1991, 4, 270; (f) Ravichandran, K.; Kerdesky, F. A. J.; Cava, M. J. Org.
Chem. 1986, 51, 2044; (g) Rho, Y. S.; Kim, S. Y.; Cho, I.; Kang, H. S.; Yoo, D. J.;
Cheong, C. Bull. Korean Chem. Soc. 1998, 19, 1059; (h) Eagon, S.; Kim, J.; Yan, K.;
Haddenham, D.; Singaram, B. Tetrahedron Lett. 2007, 48, 9025; (i) Cowden, C. J.;
Paterson, I. Org. React. 1997, 51, 38.
2
1
1
4
.4.10. Thiophen-3-ylboronic acid (7h) . Off-white solid, H NMR
13
(
(
500 MHz, MeOH-d
125 MHz, MeOH-d
þ27.0.
4
) d 7.28 (s, 1H) 7.36 (s, 1H) 7.84 (s, 1H); C NMR
4
) d 124.6,127.3,131.6,134.3; B NMR (160 MHz,
11
4
MeOH-d ) d
.4.11. 3,4-Dimethylphenylboronic acid (9d). Off-white solid, 1
4
H
NMR (500 MHz, CDCl
s, 2H); 13C NMR (125 MHz, CDCl
) d
3
3
)
d
2.61 (s, 3H), 2.65 (s, 3H), 7.54 (s, 1H), 8.24
20.0, 20.5, 128.2, 129.7, 133.7,
þ29.3; EM (ESI): m/
8 2
H11BNaO 173.07764, found 173.07443.
2. Boronic Acids; Hall, D. G., Ed.; Wiley-VCH: Germany, 2005.
3
. (a) Myung, J.; Kim, K. B.; Crews, C. M. Med. Res. Rev. 2001, 21, 245; (b) Soloway, A.
H.; Tjarks, W.; Barnum, B. A.; Rong, F. G.; Barth, R. F.; Codogni, I. M.; Wilson, J. G.
Chem. Rev. 1998, 98, 1515; (c) Kitano, S.; Koyama, Y.; Kataoka, K.; Okano, T.;
Sakurai, Y. J. Controlled Release 1992,19,161; (d) Riggs, J. A.; Hossler, K. A.; Smith, B.
D.; Karpa, M. J.; Griffin, G.; Duggan, P. J. Tetrahedron Lett.1996, 37, 6303; (e) James,
T. D.; Sandanayake, K. R. A. S.; Shinkai, S. Angew. Chem., Int. Ed. 1996, 35, 1911; (f)
Sugasaki, A.; Sugiyasu, K.; Ikeda, M.; Takeuchi, M.; Shinkai, S. J. Am. Chem. Soc.
(
1
11
36.3, 137.0, 141.8; B NMR (160 MHz, MeOH)
d
z (Mþ) calcd for C
2
1
1
4
.4.12. 4-Chlorophenylboronic acid (9f) . Off-white solid, H NMR
13
(
500 MHz, MeOD)
d
5.70 (d, 2H, J¼7.0 Hz), 6.04 (d, 2H, J¼6.5 Hz);
C
2001, 123, 10239; (g) Yang, W. Q.; Gao, X. M.; Karnati, V. V. R.; Ni, W. J.; Wang, B.;
1
1
NMR (125 MHz, MeOD)
d
d
125.7, 133.4; B NMR (160 MHz, MeOD)
Hooks, W. B.; Carson, J.; Weston, B. Bioorg. Med. Chem. Lett. 2002, 12, 2175.
. (a) Brown, H. C.; Cole, T. E. Organometallics 1983, 2, 1316; (b) Brown, H. C.; Bhat,
N. G.; Srebnik, M. Tetrahedron Lett. 1988, 29, 2631; (c) Brown, H. C.; Rangaish-
envi, M. V. Tetrahedron Lett. 1990, 49, 7113; (d) Matteson, D. S.; Liedtke, J. D.
J. Am. Chem. Soc. 1965, 87, 1526.
5. (a) Khotinsky, E.; Melamed, M. Ber. 1909, 54, 2784; (b) Gilman, H.; Vernon, C.
J. Am. Chem. Soc. 1926, 48, 1063.
. (a) Ishiyama, T.; Miyaura, N. Chem. Rec. 2004, 3, 271; (b) Ishiyama, T.; Murata,
M.; Miyaura, N. J. Org. Chem. 1995, 60, 7508.
4
þ30.4; EM (ESI): m/z (Mþ) calcd for C
6
H
11BClO
2
185.04943, found
185.05351.
2
1
1
4
.4.13. 4-Cyanophenylboronic acid (9h) . Off-white solid, H NMR
13
(
500 MHz, MeOD)
d
7.04 (d, 2H, J¼8.0 Hz), 7.23 (d, 2H, J¼8.5 Hz);
C
6
11
NMR (125 MHz, MeOD)
d
116.8, 129.0, 132.4; B NMR (160 MHz,

 Haddenham, Dustin
Haddenham, Dustin
 Bailey, Christopher L.
Bailey, Christopher L.
 Vu, Chau
Vu, Chau
 Nepomuceno, Gabby
Nepomuceno, Gabby
 Eagon, Scott
Eagon, Scott
 Pasumansky, Lubov
Pasumansky, Lubov
 Singaram, Bakthan
Singaram, Bakthan