J. Liu et al.
industrially manufactured by glyoxal oxidation with nitric
acid. However, this process produces large amounts of nitro-
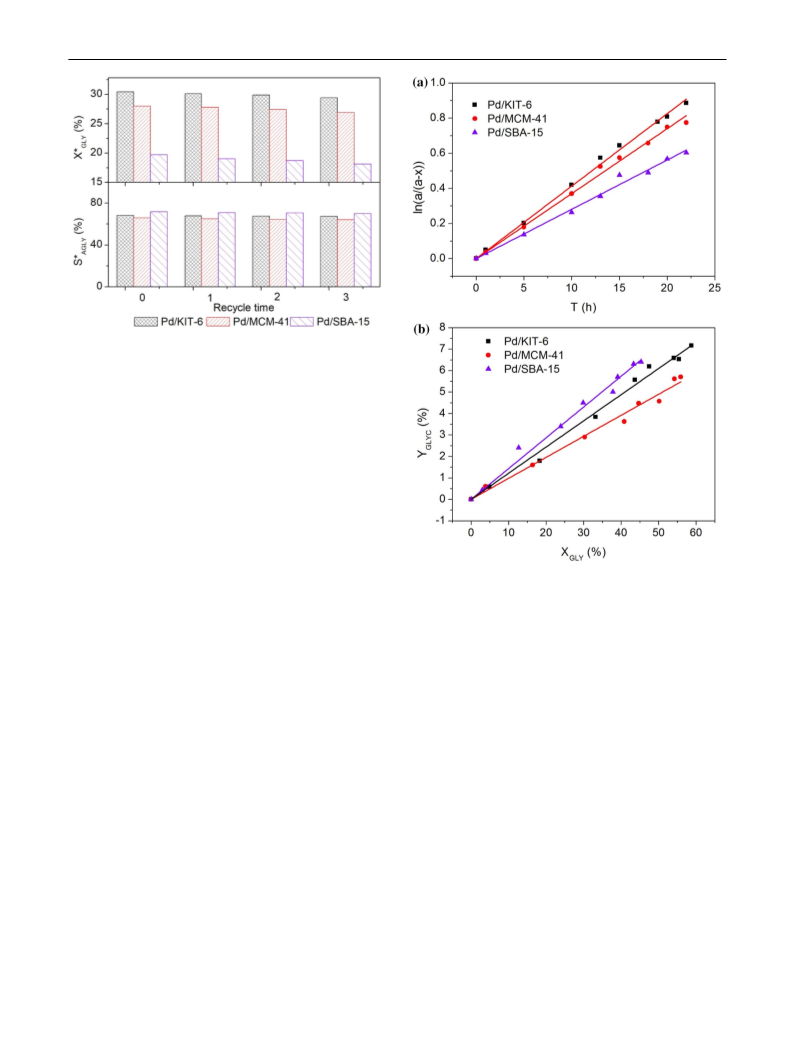
mesopore favored the mass transfer of reactants, which
helped to reduce the chance of over-oxidation. Consequently,
the dimensionality eꢃect of supports on the catalytic activity
and selectivity was investigated.
gen oxides (NO ), which causes severe potential environ-
x
mental problems. It is therefore urgent to be substituted by
green synthesis route. Direct selective oxidation of glyoxal
to glyoxalic acid, using air as oxidant, is an attractive and
environmentally-friendly route to solve these problems.
Recently, some carbon supported noble metal catalysts, such
as Pd/C, Pt/C and Au/C have been applied in the title reac-
tion as active components [4–7]. Among them, Pd/C exhib-
ited superior activity for selective oxidation of glyoxal under
mild condition. However, there are still some challenges for
Pd/C in this reaction. First, the most active monometallic
Pd/C catalyst exhibits relative low activity with about 15%
yield of glyoxalic acid after 20 h. By bimetallic modiꢀca-
tion, the Ru–Pd/C catalyst can reach around 20% yield of
glyoxalic acid [4, 8, 9]. Besides, the product, glyoxalic acid,
is easily over-oxidized to oxalic acid, which leads to low
selectivity [10]. Therefore, there is a signiꢀcant demand for
development of new catalyst systems to achieve higher activ-
ity and selectivity.
2 Experimental Sections
2.1 Synthesis of MS Supports
Mesoporous KIT-6, MCM-41 and SBA-15 were synthesized
according to the literature [20, 22]. For KIT-6, 4.0 g Pluronic
P123 was dissolved in 144 g water, 4.0 g Butan-1-ol and
7.9 g (35 wt%) hydrochloric acid with stirring at 40 °C. Then
8.6 g tetraethyl orthosilicate was added dropwise and stirred
for 24 h. Subsequently, the mixture was aged at 100 °C for
24 h without stirring. The obtained suspension was ꢀltered
and washed with water, dried overnight before calcination at
550 °C for 6 h. For MCM-41, 4.0 g hexadecyltrimethylam-
monium bromide was dissolved in 200 g water and 15.9 g
(25 wt%) aqueous ammonia with stirring at 25 °C. Then
16.7 g of tetraethyl orthosilicate was added dropwise and
stirred for 1 h. The obtained suspension was ꢀltered, washed
with water and dried overnight before calcination at 550 °C
for 6 h. For SBA-15, 4.0 g Pluronic P123 was dissolved in
127 g water and 85 g of 4 M hydrochloric acid with stirring
at 35 °C. Then 11.3 g of tetraethyl orthosilicate was added
dropwise and stirred for 20 h. The obtained suspension was
ꢀltered and washed with water, dried overnight before cal-
cination at 550 °C for 6 h samples.
Mesoporous silica (MS), as a kind of excellent catalytic
supports with high surface area, ordered pore structure and
ꢂexible pore architecture [11, 12], is widely used in the het-
erogeneous catalysis [13–15]. In the past decades, various
mesoporous silicas with tunable pore size have been synthe-
sized, such as M41S family, SBA series and MSU-X. Their
superior catalytic performance in photocatalysis reaction,
selective oxidation reaction and organic synthesis was veri-
ꢀ
ed [16–18]. Karimi et al. [19] reported Pd immobilized on
SBA-15 for the aerobic oxidation of alcohols. The Pd nano-
particles were well conꢀned inside the channel of SBA-15
and they acted as active species for the aerobic oxidation of
a wide range of alcohols. Afterwards, Parlett et al. [20, 21]
found that Pd dispersed on SBA-16 and KIT-6 exhibited
enhanced aerobic alcohol oxidation performance owing to
their three-dimensional (3D) interconnected architecture.
For the aerobic alcohol oxidation, the 3D architecture of
MS supports led to the obvious enhancement of incorporated
metal dispersion and mass transfer in liquid phase. Since
these investigated oxidation reactions did not face the prob-
lem of severe over-oxidation, their work mainly focused on
the promotion of conversion by the 3D mesoporous structure
of supports.
2.2 Preparation of Pd/MS Catalysts
The Pd (1 wt%)/MS catalysts were prepared by impregnation
method in acetic solution. 0.011 g Pd(OAc) was dissolved
2
in 50 mL acetic solution. The synthesized MS support
(0.50 g) was then added to the above solution and stirred
for 4 h at 50 °C. After that, the solvent in suspension solu-
tion was vaporized under a vacuum in a rotary evaporator.
The obtained precursor was then dried at 90 °C overnight.
Finally, the dried powder was calcined in air at 350 °C for
3 h, followed by reduction at 350 °C for 3 h in hydrogen.
These catalysts were denoted as Pd/SBA-15, Pd/MCM-41
and Pd/KIT-6.
Despite these excellent features of MS, to our knowledge,
there is no report on MS supported Pd catalyst for selective
oxidation of glyoxal to glyoxalic acid by air. Herein, we pre-
pared a series of Pd/MS catalysts (Pd/KIT-6, Pd/MCM-41
and Pd/SBA-15) to investigate their catalytic performance.
Compared with 2D architecture of MCM-41 and SBA-15,
we speculated that the 3D architecture of KIT-6 could pro-
vide highly dispersed active sites, which were more acces-
sible to reactant molecules. Meanwhile, its interconnected
2.3 Characterization
Nitrogen adsorption and desorption isotherms were per-
formed by Micromeritics ASAP2020. Samples were
degassed at 200 °C before measurements. The speciꢀc sur-
face areas were calculated by the BET equation. The pore
diameters and volumes were determined by applying the
1
3

 Liu, Junchi
Liu, Junchi
 Qin, Feng
Qin, Feng
 Huang, Zhen
Huang, Zhen
 Huang, Liang
Huang, Liang
 Liao, Zhenan
Liao, Zhenan
 Xu, Hualong
Xu, Hualong
 Shen, Wei
Shen, Wei