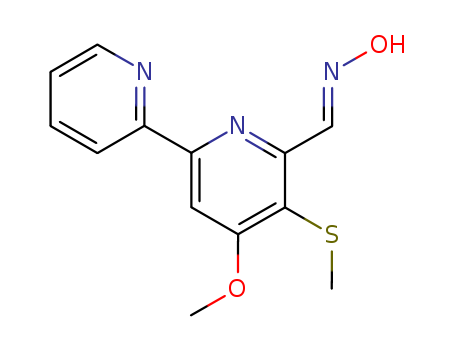
- Chemical Name:COLLISMYCIN A
- CAS No.:158792-24-6
- Molecular Formula:C13H13N3O2S
- Molecular Weight:275.331
- Hs Code.:2933399090
- Mol file:158792-24-6.mol
Synonyms:[2,2'-Bipyridine]-6-carboxaldehyde,4-methoxy-5-(methylthio)-, oxime, (E)- (9CI); Collismycin A; SF 2738A