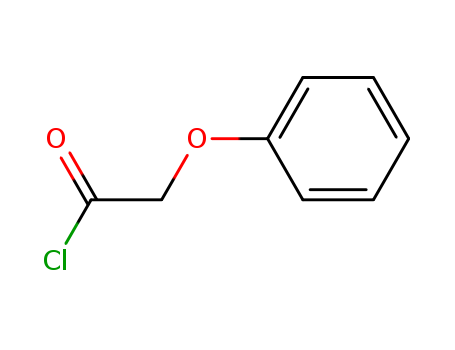
- Chemical Name:Phenoxyacetyl chloride
- CAS No.:701-99-5
- Deprecated CAS:343594-49-0
- Molecular Formula:C8H7ClO2
- Molecular Weight:170.595
- Hs Code.:29189090
- European Community (EC) Number:211-862-4
- NSC Number:9808
- UNII:NS65CKB849
- DSSTox Substance ID:DTXSID1061029
- Nikkaji Number:J62.847F
- Wikidata:Q63398649
- Mol file:701-99-5.mol
Synonyms:Phenoxyacetyl chloride;701-99-5;2-phenoxyacetyl chloride;Acetyl chloride, phenoxy-;Phenyloxyacetyl chloride;phenoxyacetic acid chloride;Acetyl chloride, 2-phenoxy-;NSC 9808;NSC-9808;EINECS 211-862-4;Phenoxyacetic acid chloride;Phenoxyethanoyl chloride;phenoxylacetylchloride;MFCD00000726;2-phenoxyacetylchloride;phenoxy acetyl chloride;phenoxy-acetyl chloride;(phenyloxy)acetyl chloride;SCHEMBL6013;Phenoxyacetyl chloride, 98%;NS65CKB849;DTXSID1061029;NSC9808;STR07065;AKOS000104374;R(+)-SKF-38393HYDROCHLORIDE;AMY202100119;FT-0632844;P0113;EN300-20022;E78941;Q63398649;F2190-0093


 C
C




