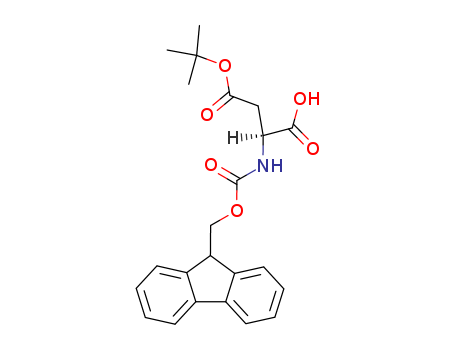
- Chemical Name:Fmoc-Asp(OtBu)-OH
- CAS No.:71989-14-5
- Molecular Formula:C23H25NO6
- Molecular Weight:411.455
- Hs Code.:29242990
- European Community (EC) Number:276-251-7
- UNII:JHQ6MY724T
- DSSTox Substance ID:DTXSID001315581
- Nikkaji Number:J295.106A
- Wikidata:Q76145921
- ChEMBL ID:CHEMBL4633466
- Mol file:71989-14-5.mol
Synonyms:Fmoc-Asp(OtBu)-OH;71989-14-5;Fmoc-L-Aspartic acid beta-tert-butyl ester;(S)-2-((((9H-Fluoren-9-yl)methoxy)carbonyl)amino)-4-(tert-butoxy)-4-oxobutanoic acid;Fmoc-Asp(OBut);Fmoc-L-Asp(OtBu)-OH;Fmoc-Asp(Ot-Bu)-OH;Fmoc-L-aspartic acid 4-tert-butyl ester;MFCD00037131;Fmoc-L-Asp(tBu);FmocAsp(OtBu)-OH;Fmoc-Asp(Obu)-OH;Fmoc-Asp(tBu)-OH;Fmoc-Asp(t-Bu)-OH;N-Alpha-Fmoc-L-aspartic acid beta-t-butyl ester;(2S)-2-(9H-fluoren-9-ylmethoxycarbonylamino)-4-[(2-methylpropan-2-yl)oxy]-4-oxobutanoic acid;4-(tert-butoxy)-2-{[(9H-fluoren-9-ylmethoxy)carbonyl]amino}-4-oxobutanoic acid;4-tert-Butyl N-[(9H-Fluoren-9-ylmethoxy)carbonyl]-L-aspartate;FmocAsp(OBut)OH;N-alpha-FMOC-L-aspartic acid beta-tert-butyl ester;L-Fmoc-asparticacidalpha-t-butylester;Fmoc-L-aspartic acid beta-tert.buty ester;ST50307352;(2S)-4-(Tert-Butoxy)-2-{[(9H-Fluoren-9-Yl-Methoxy)Carbonyl]Amino}-4-Oxobutanoic Acid;JHQ6MY724T;SCHEMBL872021;CHEMBL4633466;4-tert-Butyl N-Fmoc-L-aspartate;FODJWPHPWBKDON-IBGZPJMESA-N;DTXSID001315581;BCP08108;CS-D1469;HY-Y1169;STL466193;AKOS015924105;AC-1156;(2S)-4-(tert-butoxy)-2-({[(9H-fluoren-9-yl)methoxy]carbonyl}amino)-4-oxobutanoic acid;(2S)-4-(tert-butoxy)-2-{[(9H-fluoren-9-ylmethoxy)carbonyl]amino}-4-oxobutanoic acid;L-aspartic acid, N-[(9H-fluoren-9-ylmethoxy)carbonyl]-, 4-(1,1-dimethylethyl) ester;Fmoc-3-(tert-Butoxycarbonyl)-L-Ala-OH;Fmoc-L-aspartic acid b-tert-butyl ester;AS-14174;BP-23513;BP-30008;N-Fmoc-L-aspartic Acid 4-tert-Butyl Ester;AM20060822;B3150;Fmoc-Asp(OtBu)-OH, >=98.0% (HPLC);EN300-96401;M03404;N-?-FMOC-L-aspartic acid ?-tert-butyl ester;A837378;J-300058;4-tert-Butyl N-(fluoren-9-ylmethoxycarbonyl)-L-aspartate;N-(Fluoren-9-ylmethoxycarbonyl)-L-aspartic acid 4-tert-butyl ester;N-9-Fluorenylmethyloxycarbonyl-L-aspartic acid beta-t-butyl ester;N-[(9H-Fluoren-9-ylmethoxy)carbonyl]-L-aspartic Acid 4-tert-Butyl Ester;(s)-2-(((9h-fluoren-9-yl)methoxy)carbonylamino)-4-tert-butoxy-4-oxobutanoic acid;(2S)-4-tert-butoxy-2-{[(9H-fluoren-9-ylmethoxy)carbonyl]amino}-4-oxobutanoic acid (non-preferred name);N-alpha-(9-Fluorenylmethyloxycarbonyl)-L-aspartic acid beta-t-butyl ester (Fmoc-L-Asp(OtBu)-OH)


 Xi
Xi




