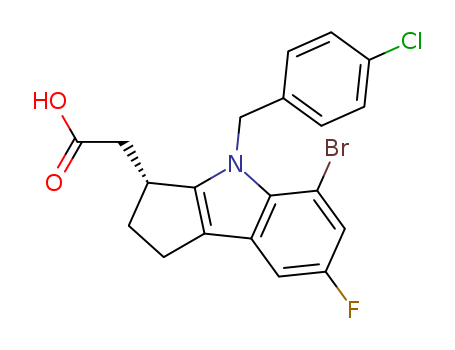
- Chemical Name:(R)-2-(5-broMo-4-(4-chlorobenzyl)-7-fluoro-1,2,3,4-tetrahydrocyclopenta[b]indol-3-yl)acetic acid
- CAS No.:393509-05-2
- Molecular Formula:C20H16BrClFNO2
- Molecular Weight:436.708
- Hs Code.:
Synonyms:(R)-2-(5-broMo-4-(4-chlorobenzyl)-7-fluoro-1,2,3,4-tetrahydrocyclopenta[b]indol-3-yl)acetic acid