10.1016/S0040-4020(01)89811-2
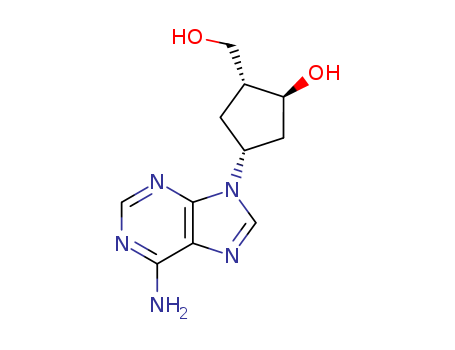
This study presents an improved method for the synthesis of (+)-carbocyclic 2'-deoxyadenosine, a nucleoside analog with potential antiviral properties, starting from the bicyclic lactone diol (+)-1. The key steps involve the sequential iodide decarboxylation of the carboxylic acid side chain derived from the lactone moiety of (+)-1 using iodobenzene diacetate (IBDA) and a hypervalent iodine species. The tetrahydropyranyl (THP) group was used for protection, replacing the secondary mesyloxy functionality with an azide anion, while a complete inversion of the configuration was performed. The heterocyclic moiety (adenine) was constructed by a modified literature method involving the reduction of NO2 to NH2 groups with SnCl2. The study also reports an unexpected formation of a disubstituted 2-oxabicyclo[2.2.1]heptane skeleton formed via a hypervalent iodine species derived from the intermediate. The overall yield and purity of the final product were improved, highlighting the efficiency of the synthetic route and the importance of the reagents and conditions used.