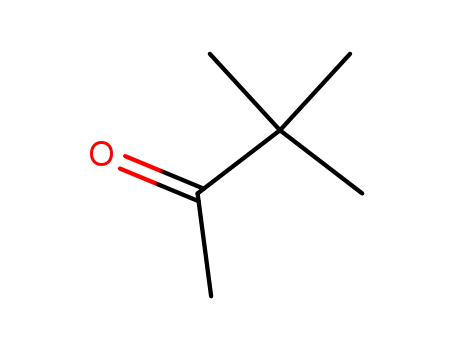
- Chemical Name:3,3-Dimethyl-2-butanone
- CAS No.:75-97-8
- Molecular Formula:C6H12O
- Molecular Weight:100.161
- Hs Code.:29141990
- European Community (EC) Number:200-920-4
- NSC Number:935
- UNII:3U1AAG3528
- DSSTox Substance ID:DTXSID5021752
- Nikkaji Number:J4.165C
- Wikipedia:Pinacolone
- Wikidata:Q222983
- ChEMBL ID:CHEMBL3183565
- Mol file:75-97-8.mol
Synonyms:3,3-Dimethyl-2-butanone;Pinakolin [German];2-Butanone,3,3-dimethyl-;3,3-Dimethylbutanone;t-butyl methyl ketone;Pinacoline;Methyl t-butyl ketone;3,3-dimethylbutan-2-one;tert-Butyl methyl ketone;2, 2-Dimethyl-3-butanone;Ketone, tert-butyl methyl;2,2-Dimethyl-3-butanone;Methyltert-butyl ketone;2-Butanone, 3,3-dimethyl-;4-01-00-03310 (Beilstein Handbook Reference);1,1,1-Trimethylacetone;2,2-Dimethylbutanone;3, 3-Dimethyl-2-butanone;Ketone, t-butyl methyl;Pinacolin;Pinakolin;Methyl tert-butyl ketone;


 F,
F,  Xn,
Xn,  Xi
Xi
 F:Flammable;
F:Flammable;



