10.1021/ac034876a
The study presents the development of a biosensor-based assay for the quantification of riboflavin (Rf) in milk samples using surface plasmon resonance (SPR) technology. The assay involves the indirect measurement of Rf by detecting the excess of riboflavin binding protein (RBP) that remains free after complexation with Rf molecules originally present in the sample. The sensor chip is modified with covalently immobilized Rf to bind the excess RBP. The method involves a chemical modification to introduce a reactive ester group on the Rf molecule for immobilization on the chip surface. Calibration solutions are prepared by mixing Rf standard solutions with an optimized concentration of RBP, and the Rf content in milk samples is measured by comparing the response against the calibration. The results are comparable to those obtained from an official HPLC-fluorescence procedure, with a limit of quantification determined to be 234 μg/L and a limit of detection to 70 μg/L. The study demonstrates the potential of SPR-based biosensors as a competitive alternative to traditional analytical techniques for the determination of riboflavin in food samples.
10.1016/j.tet.2005.05.055
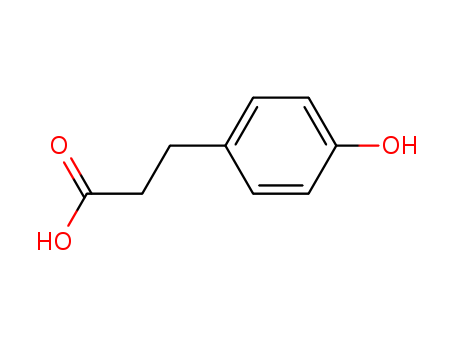
The study focuses on the synthesis and evaluation of galanthamine analogs, specifically 6H-benzofuro[3a,3,2-e,f][1]benzazepine and 6H-benzofuro[3a,3,2-e,f][3]benzazepine, which are derivatives of the Amarylidaceae alkaloid galanthamine. Galanthamine is known for its cholinesterase inhibitory properties and is used in the treatment of neuromuscular diseases and Alzheimer's dementia. The purpose of the study was to alter the position of the nitrogen within the azepine ring of galanthamine to create analogs that might have similar or improved therapeutic effects. The synthesis involved a variety of chemicals, including p-hydroxyphenylpropionic acid, benzyl chloride, thionyl chloride, and various other reagents and solvents, which were used to perform esterification, benzylation, saponification, formylation, bromination, and reduction reactions, among others. These chemicals served to construct the complex molecular structures of the analogs, with the ultimate goal of understanding how changes in the molecular structure affect the reactivity and potential therapeutic applications of these compounds.


 Xi
Xi



