10.1021/ic00149a018
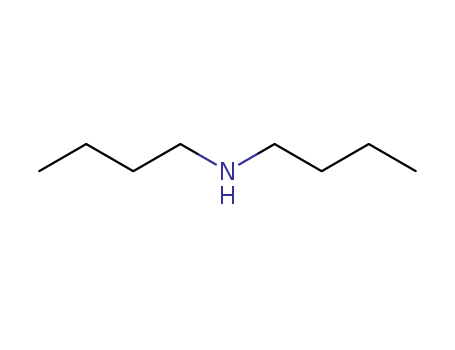
The research focused on the kinetics and mechanisms of dealkylation reactions of N-methylporphyrin complexes, specifically examining the effects of porphyrin ring substituents and reaction media on these reactions. The study aimed to understand the dealkylation process of copper(II) complexes of N-methylporphyrins with various nucleophiles under mild conditions, which is relevant to the formation of N-alkylporphyrins from the interaction of cytochrome P-450 with substrates and has implications for the synthesis of metal complexes used in medical applications. The researchers found that the reactions were first order with respect to the copper(II) N-methylporphyrin complexes and nucleophile concentration at low nucleophile concentrations, with a negative deviation from linearity at high nucleophile concentrations, suggesting a mechanism involving two first-order paths. They concluded that even large differences in ring substituents have little effect on the rates of dealkylation promoted by metal ions, and the reaction medium can significantly influence the rate and mechanism of the reaction. Key chemicals used in the study included copper(II) complexes of N-methyltetraphenylporphyrin, N-methyltetrakis(psulfopheny1)porphyrin, and N-methyldeuteroporphyrin IX dimethyl ester, along with nucleophiles such as di-n-butylamine, diethylamine, pyridine, and chloride ion in solvents like acetonitrile, water, and dichloromethane.
10.1021/ja01644a034
The study investigates the synthesis of various formamidines and their derivatives using different chemical reactions and methods. Key chemicals involved include formic acid, various amines (such as n-octylamine, di-n-butylamine, and aniline), phosphorus pentachloride, and formanilides. These chemicals play crucial roles in the formation of formamidines through condensation reactions, interactions in the presence of chlorinating agents, and other synthetic processes. The study explores the preparation of disubstituted, heterodisubstituted, and trisubstituted formamidines, as well as their hydrochlorides and other derivatives. The research also examines the stability, hydrolysis, and qualitative testing of these compounds, providing insights into their chemical properties and potential applications.


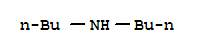
 Xn
Xn
 Xn:Harmful;
Xn:Harmful;



