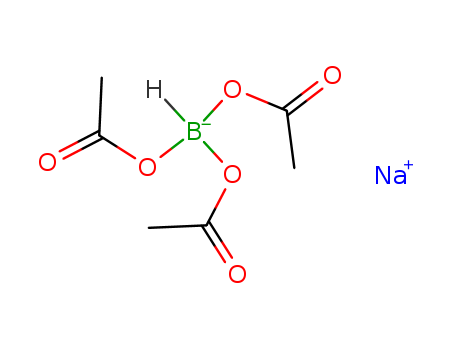
- Chemical Name:Sodium triacetoxy-borohydride
- CAS No.:56553-60-7
- Molecular Formula:C6H10BNaO6
- Molecular Weight:211.943
- Hs Code.:2850.00
- Nikkaji Number:J1.884.353F,J2.805.651F
- Wikidata:Q7553383
- Mol file:56553-60-7.mol
Synonyms:sodium trisacetoxyborohydride;sodium triacetoxy-borohydride;Na(OAc)3BH;NaB(OAc)3H;NaBH(AcO)3;NaBH(OAc)3;sodium tnacetoxyborohydnde;UNII-4VU0JE4YSK;sodium tnacetoxyborohydride;sodium triacetoxyborohydnde;sodium triactoxyborohydride;sodiumtrisacetoxyborohydride;sodium triacetoxvborohydride;sodium triacetoxyhydroborate;sodium triacteoxyborohydride;sodiumtriacetoxy borohydride;sodiumtriacetoxy-borohydride;C6H10BNaO6;Sodium triacetoxiborohydride;sodium triacetoxyboro hydrid;sodium triacetoxylborohydride;sodium tri-acetoxyborohydride;sodium triacetoxyboro hydride;sodium triacetoxyboro-hydride;sodium triacetoxyborohyd ride;sodium(triacetoxy)borohydride;Sodium triacetoxy borohydride;Sodium triacetoxyl borohydride;Sodium triacetoxyl-borohydride;Sodium tris-acetoxyborohydride;sodium trisacetoxy borohydride;sodium (triacetoxy)borohydride;sodium tri acetoxy borohydride;sodium tri(acetoxy)borohydride;sodium tri-acetoxy borohydride;sodium tri-acetoxy-borohydride;sodium(triacetoxy)-borohydride;sodium triacetyloxy borohydride;Sodium tris(acetoxy)borohydride;Sodium tris(acetoxy)hydroborate;sodium (triacetoxy)-borohydride;DTXSID8074368;sodium tris(acetoxy) borohydride;HHYFEYBWNZJVFQ-UHFFFAOYSA-N;BP-31215;NS00123020;Sodium Triacetoxyborohydride (Technical Grade);D77878;Q7553383


 F,
F,  Xi,
Xi,  C
C




