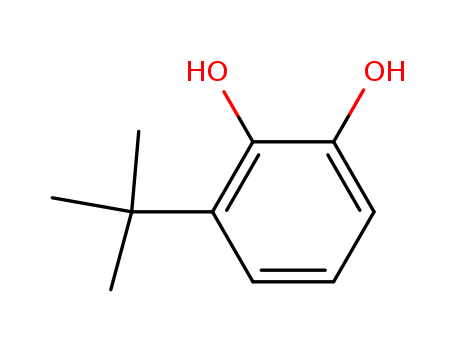
- Chemical Name:3-(tert-Butyl)benzene-1,2-diol
- CAS No.:4026-05-5
- Deprecated CAS:1319-92-2,29863-27-2
- Molecular Formula:C10H14O2
- Molecular Weight:166.22
- Hs Code.:2907299090
- European Community (EC) Number:223-695-4
- UNII:47F26P1WUE
- Nikkaji Number:J141.449F
- Wikidata:Q27259050
- Mol file:4026-05-5.mol
Synonyms:3-(tert-Butyl)benzene-1,2-diol;4026-05-5;3-tert-butylbenzene-1,2-diol;3-tert-butylcatechol;3-tert-Butylpyrocatechol;tert-Butylpyrocatechol;t-butylcatechol;MFCD00515124;t-butylpyrocatechol;tert-butyl catechol;tert.-butylcatechol;29863-27-2;SCHEMBL36109;47F26P1WUE;06J124VX0O;3-(tert-Butyl)-1,2-benzenediol;EINECS 223-695-4;PYROCATECHOL, 3-TERT-BUTYL-;CS-W006764;AS-58276;SY104770;1,2-Benzenediol, 3-(1,1-dimethylethyl)-;W17828;EN300-1599500;Q27259050