10.1002/chem.201604507
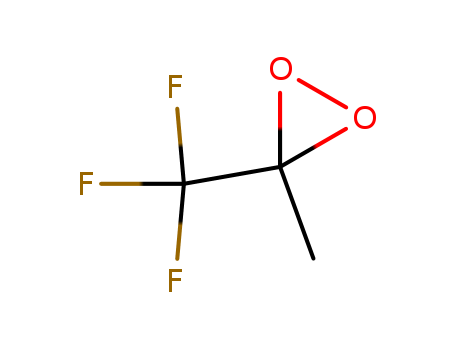
The research investigates the impact of the electronic properties of dioxiranes on the chemoselectivity of lactam oxidation, aiming to understand how changes in the oxidant's structure influence the reaction pathway. The study compares the oxidation of various lactams using dimethyl dioxirane (DDO) and methyl(trifluoromethyl)dioxirane (TFDO), revealing an unprecedented switch from C-H to N-H oxidation as the fluorine content in the dioxirane increases. The findings, supported by experimental data and computational studies, indicate that the presence of electron-withdrawing fluorine atoms in the dioxirane structure stabilizes a more polar transition state, favoring the heterolytic (2e) N-H oxidation pathway over the homolytic (1e) C-H oxidation. This work highlights the potential for tuning the selectivity of oxidation reactions by modifying the electronic characteristics of the oxidant, offering new insights into the mechanisms of dioxirane-mediated oxidations.