Products Categories
| CAS No.: | 1617-90-9 |
|---|---|
| Name: | Vincamine |
| Molecular Structure: | |
|
|
|
| Formula: | C21H26N2O3 |
| Molecular Weight: | 354.449 |
| Synonyms: | Vincapan;Pervincamine;Decincan;Eburnamenine-14-carboxylic acid,14,15-dihydro-14-hydroxy-,methyl ester,(3R,14a,16R)-;Vincamidol;Perval;Methyl vincaminate;Monorin;Eburnamenine-14-carboxylic acid, 14,15-dihydro-14-hydroxy-, methyl ester, (3alpha,14beta,16alpha)-;Minorin;Vinkametrin;Vinca-Ecobi;Anasclerol;Vincimax;Eburnamenine-14-carboxylic acid, 14,15-dihydro-14-hydroxy-, methyl ester, (3.alpha.,14.beta.,16.alpha.)-;Vinca-Minor;Equipur;Minorine; |
| EINECS: | 216-576-3 |
| Density: | 1.36 g/cm3 |
| Melting Point: | 232 °C (dec.)(lit.) |
| Boiling Point: | 508.9 °C at 760 mmHg |
| Flash Point: | 261.6 °C |
| Appearance: | white to almost white fine crystalline powder |
| Hazard Symbols: |
 Xn Xn
|
| Risk Codes: | 22 |
| Safety: | 36 |
| PSA: | 54.70000 |
| LogP: | 2.89040 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 211914-50-0N-[[2-[[[4-(Aminoiminomethyl)phenyl]amino]methyl]-1-methyl-1H-benzimidazol-5-yl]carbonyl]-N-(2-pyridinyl)-beta-alanine ethyl ester hydrochloride

- 142892-63-5
(+)-15α-Chloro-vincamine


- 1617-90-9
vincamin

| Conditions | Yield |
|---|---|
| With hydrogen; potassium carbonate; triethylamine; palladium on activated charcoal In methanol for 6h; Ambient temperature; | 84.7% |

| Conditions | Yield |
|---|---|
| at 250℃; for 0.25h; | A 7% B 83% |
| at 20℃; for 0.25h; | A 8% B 75% |

| Conditions | Yield |
|---|---|
| Stage #1: (-)-vincadifformine With monoperoxymaleic acid In methanol at -5 - 0℃; for 6.16667h; Stage #2: With sodium dithionate at 40℃; for 3h; Reagent/catalyst; | 78.3% |

| Conditions | Yield |
|---|---|
| In toluene at 450℃; under 20 Torr; | A 5% B 70% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In acetone for 72h; Ambient temperature; Yields of byproduct given; | A n/a B 66% |

- 85588-92-7
(-)-methyl 1,2,3,4,6,7,12,12bα-octahydroindolo<2,3-a>quinolizine(1β-yl) pyruvate oxime

A

- 6835-99-0
14-epivincamine

B

- 1617-90-9
vincamin

| Conditions | Yield |
|---|---|
| With sodium disulfite; acetic acid In water at 90 - 92℃; for 6h; Yields of byproduct given; | A n/a B 60% |
| With sodium disulfite; acetic acid In water at 90 - 92℃; for 6h; Yield given. Title compound not separated from byproducts; | A n/a B 60% |
| With sodium disulfite; acetic acid In water at 92 - 95℃; for 5h; | A 20% B 40% |

| Conditions | Yield |
|---|---|
| at 150℃; for 0.25h; | A 35% B 60% |
| at 150℃; for 0.25h; other condition; | A 35% B 60% |
| In acetic acid at 20℃; for 12h; | A 4 mg B 50 mg |
| In acetic acid for 12h; Ambient temperature; | A 4 mg B 50 mg |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; ammonia; sodium chloride In methanol | A n/a B 58% |


| Conditions | Yield |
|---|---|
| In toluene at 400℃; under 20 Torr; | A 9% B 47% C 10% |


| Conditions | Yield |
|---|---|
| With sodium hydroxide; oxygen; sodium acetate; rose bengal; sodium thiosulfate; acetic acid 1.) methanol, irradation, 60 min, 2.) H2O, 70 deg C, 20 min; Yield given. Multistep reaction; | A n/a B 46% |
| With sodium hydroxide; oxygen; sodium acetate; rose bengal; sodium thiosulfate; acetic acid 1.) methanol, irradation, 60 min, 2.) H2O; Yield given. Multistep reaction. Yields of byproduct given; | |
| With sulfuric acid; ozone In methanol at 60℃; Yield given. Yields of byproduct given; |
Chemistry
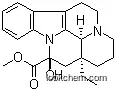
Molecular Structure of Vincamine (CAS NO.1617-90-9):
.png)
Molecular Formula: C21H26N2O3
Molecular Weight: 354.44
CAS Registry Number: 1617-90-9
H bond acceptors: 5
H bond donors: 1
Freely Rotating Bonds: 4
Polar Surface Area: 43.7 Å2
Index of Refraction: 1.681
Molar Refractivity: 98.28 cm3
Molar Volume: 259.6 cm3
Surface Tension: 54.2 dyne/cm
Density: 1.36 g/cm3
Flash Point: 261.6 °C
Enthalpy of Vaporization: 82.06 kJ/mol
Boiling Point: 508.9 °C at 760 mmHg
Vapour Pressure: 3.53E-11 mmHg at 25°C
Melting point: 232 oC (dec.)
EINECS: 216-576-3
Storage temp: 2-8°C
Merck: 9983
Product Categories: Alkaloids;Biochemistry; Indole Alkaloids; AlkaloidAsymmetric Synthesis; Biochemicals Found in Plants; Chiral Building Blocks; Complex Molecules; Nutrition Research
Toxicity Data With Reference
| 1. | ipr-rat LD50:253 mg/kg | ARZNAD Arzneimittel-Forschung. Drug Research. 32 (1982),601. | ||
| 2. | orl-mus LD50:1 g/kg | ARZNAD Arzneimittel-Forschung. Drug Research. 10 (1960),811. | ||
| 3. | ipr-mus LD50:215 mg/kg | EJMCA5 European Journal of Medicinal Chemistry. Chimie Therapeutique. 16 (1981),191. | ||
| 4. | ivn-mus LD50:47,740 µg/kg | AGSOA6 Agressologie. Revue Internationale de Physio-Biologie et de Pharmacologie Appliquees aux Effets de l’Agression. 19 (1978),211. |
Safety Profile
Poison by intravenous and intraperitoneal routes. Moderately toxic by ingestion. When heated to decomposition it emits toxic fumes of NOx. Used as a vasodilator.
Hazard Codes:  Xn
Xn
Risk Statements: 22
R22:Harmful if swallowed.
Safety Statements: 36
S36:Wear suitable protective clothing.
WGK Germany: 3
RTECS: YY8575000
Specification
Vincamine , with CAS number of 1617-90-9, can be called Eburnamenine-14-carboxylic acid, 14,15-dihydro-14-hydroxy-, methyl ester, (3alpha,14beta,16.) ; Oligo Proanthocyanidin ; Vincasaunier ; Vincadar ; 14,15-Dihydro-14-hydroxyeburnamenine-14-carboxylic acid methyl ester ; Eburnamenine-14-carboxylic acid, 14,15-dihydro-14-hydroxy-, methyl ester, (3.alpha.,14.beta.,16.alpha.)- . It is a white to almost white fine crystalline powder, Vincamine is often used as a nootropic agent to combat the effects of aging, or in conjunction with other nootropics (such as piracetam) for a variety of purposes. Vincamine (CAS NO.1617-90-9) is a peripheral vasodilator that increases blood flow to the brain. Vincamine is an indole alkaloid (specifically a tryptamine) found in the leaves of Vinca minor, comprising about 25-65% of the indole alkaloids found in Vinca minor by weight. Vincamine is also found in the species Catharanthus roseus. Vincamine can be synthesized from related alkaloids.