Products Categories
| CAS No.: | 77-67-8 |
|---|---|
| Name: | ETHOSUXIMIDE |
| Article Data: | 12 |
| Molecular Structure: | |
|
|
|
| Formula: | C7H11 N O2 |
| Molecular Weight: | 141.17 |
| Synonyms: | Succinimide,2-ethyl-2-methyl- (6CI,7CI,8CI); (?à)-Ethosuximide; 2-Ethyl-2-methylsuccinimide; 2-Methyl-2-ethylsuccinimide;3-Ethyl-3-methylpyrrolidine-2,5-dione; 3-Ethyl-3-methylsuccinimide;3-Methyl-3-ethylpyrrolidine-2,5-dione; 3-Methyl-3-ethylsuccinimide; Atysmal;C.I. 366; CN 10395; Capitus; Emeside; Epileo Petitmal; Ethosuxide;Ethosuximide; Ethymal; Etosuximid; H 940; Mesentol; NSC 64013; PM 671; Pemal;Peptinimid; Petinimid; Petnidan; Piknolepsin; Pyknolepsinum; Ronton; Simatin;Succimal; Succimitin; Suxilep; Suximal; Suxinutin; Zarodan; Zarondan;Zarondan-Saft; Zarontin; a-Ethyl-a-methylsuccinimide;a-Methyl-a-ethylsuccinimide |
| EINECS: | 201-048-7 |
| Density: | 1.1522 (rough estimate) |
| Melting Point: | 51 °C |
| Boiling Point: | 265.3 °C at 760 mmHg |
| Flash Point: | 123.8 °C |
| Solubility: | ethanol: 100 mg/mL |
| Hazard Symbols: |
 Xn Xn
|
| Risk Codes: | 22 |
| Safety: | 36 |
| PSA: | 46.17000 |
| LogP: | 0.77800 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- 90221-55-92-bromo-5-methylbenzaldehyde
- 885590-99-82,3-DIFLUORO-4-IODOBENZALDEHYDE
- 97730-31-9(S)-4'-(2-Methylbutyl)Biphenyl-4-Carbonitrile

- 77-67-8
ethosuximide

| Conditions | Yield |
|---|---|
| Stage #1: C14H17NO3 With palladium on activated charcoal; hydrogen Stage #2: With triethylamine; α-bromoacetophenone | 83% |
- 166324-36-3
4-ethyl-1-(4-methoxybenzyl)-4-methylpyrrolidine-2,5-dione

- 77-67-8
ethosuximide

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate In water; acetonitrile for 1h; Ambient temperature; | 47% |

| Conditions | Yield |
|---|---|
| With ammonia | |
| With ammonium hydroxide at 200℃; | |
| With ammonium hydroxide In water at 190℃; for 1.5h; |

- 77-67-8
ethosuximide

| Conditions | Yield |
|---|---|
| durch Destillation; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 1.) MgSO4, 2.) N,N-diethylaniline / 1.) Et2O, 2 h, RT, 2.) toluene, -78 deg C to RT, 24 h 2: N-chlorosuccinimide / 40 deg C, 2 h; 80 deg C, 1 h 3: SiO2, H2O / 1.) 35 deg C, overnight then 60 deg C, 30 min, 2.) benzene, 1 h 4: 58 percent / Raney nickel (W-2) / ethanol / 3 h / Heating 5: 56 percent / pyridinium chlorochromate / CH2Cl2 / 2 h / Ambient temperature 6: 47 percent / cerium(IV) ammonium nitrate / acetonitrile; H2O / 1 h / Ambient temperature View Scheme |
- 166324-35-2
4-ethyl-5-hydroxy-1-(4-methoxybenzyl)-4-methylpyrrolidin-2-one

- 77-67-8
ethosuximide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 56 percent / pyridinium chlorochromate / CH2Cl2 / 2 h / Ambient temperature 2: 47 percent / cerium(IV) ammonium nitrate / acetonitrile; H2O / 1 h / Ambient temperature View Scheme |
- 166324-34-1
4-ethyl-5-hydroxy-1-(4-methoxybenzyl)-4-methyl-3-(methylthio)pyrrolidin-2-one

- 77-67-8
ethosuximide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 58 percent / Raney nickel (W-2) / ethanol / 3 h / Heating 2: 56 percent / pyridinium chlorochromate / CH2Cl2 / 2 h / Ambient temperature 3: 47 percent / cerium(IV) ammonium nitrate / acetonitrile; H2O / 1 h / Ambient temperature View Scheme |

- 77-67-8
ethosuximide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: N-chlorosuccinimide / 40 deg C, 2 h; 80 deg C, 1 h 2: SiO2, H2O / 1.) 35 deg C, overnight then 60 deg C, 30 min, 2.) benzene, 1 h 3: 58 percent / Raney nickel (W-2) / ethanol / 3 h / Heating 4: 56 percent / pyridinium chlorochromate / CH2Cl2 / 2 h / Ambient temperature 5: 47 percent / cerium(IV) ammonium nitrate / acetonitrile; H2O / 1 h / Ambient temperature View Scheme |
- 1027595-29-4
2-Chloro-N-(4-methoxy-benzyl)-N-((E)-2-methyl-but-1-enyl)-2-methylsulfanyl-acetamide

- 77-67-8
ethosuximide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: SiO2, H2O / 1.) 35 deg C, overnight then 60 deg C, 30 min, 2.) benzene, 1 h 2: 58 percent / Raney nickel (W-2) / ethanol / 3 h / Heating 3: 56 percent / pyridinium chlorochromate / CH2Cl2 / 2 h / Ambient temperature 4: 47 percent / cerium(IV) ammonium nitrate / acetonitrile; H2O / 1 h / Ambient temperature View Scheme |

- 77-67-8
ethosuximide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: copper (I) acetate; Bathocuproine; oxygen / toluene / 80 °C / 760.05 Torr 2: hydrogen; palladium on activated charcoal View Scheme |
Chemistry
Molecular Structure of Ethosuximide (CAS NO.77-67-8):
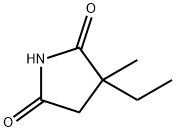
EINECS: 201-048-7
IUPAC Name: 3-Ethyl-3-methylpyrrolidine-2,5-dione
Molecular Formula: C7H11NO2
Molecular Weight: 141.167740 g/mol
XLogP3: 0.4
H-Bond Donor: 1
H-Bond Acceptor: 2
Canonical SMILES: CCC1(CC(=O)NC1=O)C
InChI: InChI=1S/C7H11NO2/c1-3-7(2)4-5(9)8-6(7)10/h3-4H2,1-2H3,(H,8,9,10)
InChIKey: HAPOVYFOVVWLRS-UHFFFAOYSA-N
Index of Refraction: 1.608
Molar Refractivity: 35.06 cm3
Molar Volume: 101.3 cm3
Surface Tension: 56.1 dyne/cm
Density: 1.274 g/cm3
Flash Point: 77.9 °C
Melting Point: 51 °C
Enthalpy of Vaporization: 44.14 kJ/mol
Boiling Point: 205.2 °C at 760 mmHg
Vapour Pressure: 0.254 mmHg at 25 °C
Water Solubility: 5.51e+004 mg/L at 25 °C
BRN: 0117054
Toxicity Data With Reference
| 1. | cyt-hmn:lyms 30 mg/L | MUREAV Mutation Research. 204 (1988),623. | ||
| 2. | orl-rat TDLo:66,600 µg/kg (6-14D preg):REP | 40YJAX Evaluation of Embryotoxicity, Mutagenicity and Carcinogenicity Risks In New Drugs Benesova, O., et al., eds.,Prague, Czechoslovakia.: Univerzita Karlova,1970,59. | ||
| 3. | orl-mus LD50:1530 mg/kg | EPILAK Epilepsia. 4 (4)(1963),66. | ||
| 4. | ipr-mus LD50:1752 mg/kg | EPILAK Epilepsia. 29 (1988),198. | ||
| 5. | scu-mus LD50:1810 mg/kg | NIIRDN “Drugs in Japan. Ethical Drugs, 6th Edition 1982“ Edited by Japan Pharmaceutical Information Center. 6 (1982),117. | ||
| 6. | ivn-mus LD50:780 mg/kg | NEPHBW Neuropharmacology. 24 (1985),427. |
Safety Profile
Safety Information of Ethosuximide (CAS NO.77-67-8):
Hazard Codes: Xn 
Risk Statements: 22
R22:Harmful if swallowed
Safety Statements: 36
S36:Wear suitable protective clothing
WGK Germany: 3
RTECS: WN2800000
Moderately toxic by ingestion, intravenous, subcutaneous, and intraperitoneal routes. Experimental teratogenic and reproductive effects. Mutation data reported. An anticonvulsant. When heated to decomposition it emits toxic fumes of NOx.
Specification
Ethosuximide with CAS registry number of 77-67-8 is also known as Ethosuximide [USAN:INN:BAN:JAN] ; (+-)-2-Ethyl-2-methylsuccinimide ; 2,5-Pyrrolidinedione, 3-ethyl-3-methyl- ; 2,5-Pyrrolidinedione, 3-ethyl-3-methyl-, (+-)- ; 2-Ethyl-2-methylsuccinimide ; 2-Methyl-2-ethylsuccinimide ; 3-Ethyl-3-methylpyrrolidine-2,5-dione ; 3-Ethyl-3-methylsuccinimide ; 3-Methyl-3-ethylpyrrolidine-2,5-dione ; 3-Methyl-3-ethylsuccinimide ; 5-21-09-00595 (Beilstein Handbook Reference) ; Aethosuccimidum ; Aethosuximide ; Aethosuximide [German] ; Asamid ; Atysmal ; C.I. 366 ; CI 366 ; CN-10,395 ; Capitus ; Emeside ; Epileo Petit MAL ; Ethosuccimide ; Ethosuccinimide ; Ethosuxide ; Ethosuximide ; Ethosuximidum ; Ethosuximidum [INN-Latin] ; Ethymal ; Etomal ; Etosuccimide ; Etosuccimide [DCIT] ; Etosuximid ; Etosuximida ; Etosuximida [INN-Spanish] ; Etosuximide ; H 940 ; H-490 ; HSDB 1119 ; Mesentol ; N-Ethyl methylsuccinimide ; NSC 64013 ; PM 671 ; Pemal ; Pemalin ; Pentinimid ; Peptinimid ; Petinimid ; Petnidan ; Piknolepsin ; Pyknolepsinum ; Ronton ; Simatin ; Simatin(E) ; Succimal ; Succimitin ; Succinimide, 2-ethyl-2-methyl- ; Suxilep ; Suximal ; Suxin ; Suxinutin ; Thetamid ; Thilopemal ; UNII-5SEH9X1D1D ; Zaraondan ; Zarodan ; Zarondan ; Zarondan-saft ; Zarontin ; Zartalin ; alpha-Ethyl-alpha-methylsuccinimide ; alpha-Methyl-alpha-ethylsuccinimide ; gamma-Methyl-gamma-ethyl-succinimide . It is a drug for treating absence seizures under the trade names Emeside and Zarontin due to the mechanism of action which is ethosuximide is a T-type calcium channel blocker with an IC50 of 12 ± 2 mmol/L. Emeside capsules were discontinued by their manufacturer, Laboratories for Applied Biology, in 2005. Zarontin capsules were discontinued by Pfizer in 2007. Syrup preparations of both brands are still available.


