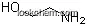Products Categories
| CAS No.: | 94421-68-8 |
|---|---|
| Name: | ANANDAMIDE |
| Molecular Structure: | |
|
|
|
| Formula: | C22H37 N O2 |
| Molecular Weight: | 347.541 |
| Synonyms: | 5,8,11,14-Eicosatetraenamide,N-(2-hydroxyethyl)-, (all-Z)-; Anandamide; Arachidonylethanolamide;N-(2-Hydroxyethyl)arachidonamide; N-(2-Hydroxyethyl)arachidonylamide;N-Arachidonoylethanolamide; N-Arachidonylethanolamide;N-Arachidonylethanolamine |
| EINECS: | 200-578-6 |
| Density: | 0.92 g/mL at 25 °C(lit.) |
| Boiling Point: | 522.3 ºCat 760 mmHg |
| Flash Point: | 269.7 ºC |
| Solubility: | 413ug/L(25 oC) |
| Appearance: | light yellow oil |
| Hazard Symbols: |
 F F
|
| Risk Codes: | 11 |
| Safety: | 24/25-16-7 |
| Transport Information: | UN 1170 3 |
| PSA: | 49.33000 |
| LogP: | 5.63150 |
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 97730-31-9(S)-4'-(2-Methylbutyl)Biphenyl-4-Carbonitrile
- 95789-13-21,3,7-trimethylpurine-2,6-dione
- 956104-42-02-Bromo-5-nitro-3-(trifluoromethyl)pyridine
- 945683-37-41-butyl-2,3-dimethylimidazolium octanesulfate
- 946052-36-41-Butyl-3-methylpyridinium methanesulfonate
- 944329-24-22-Acetamido-1,4-imino-1,2,4-trideoxy-L-arabinitol
- 960294-15-95-BROMOISOQUINOLIN-1-AMINE
- 960294-12-6tert-Butyl 2-(2-chloro-ethyl)-2-cyanopyrrolidine-1-carboxylate
- 61791-14-8Amines,coco alkyl,ethoxylated
- 61791-31-9Diethanolamine N-coco alkyl derivs.
- 7718-98-1Vanadium chloride (VCl3)
- 9003-29-6Butene, homopolymer
- 9003-06-9Poly(acrylamide-co-acrylic acid)
- 4314-63-0LYSERGIC ACID MORPHOLIDE
- 112-95-8Eicosane
- 37312-62-2Proteinase, Serratiaextracellular
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
Chemistry
Chemistry informtion about Anandamide (CAS NO.94421-68-8) is:
IUPAC Name: (5Z,8Z,11Z,14Z)-N-(2-hydroxyethyl)icosa-5,8,11,14-Tetraenamide
Synonyms: 11,14-Eicosatetraenamide(All-Z)-N-(2-Hydroxyethyl)-8 ; 14-Eicosatetraenamide,N-(2-Hydroxyethyl)-(All-Z)-11 ; 5,8,11,14-Eicosatetraenoylethanolamide ; Anandamide(20.4,N-6) ; N-(2-Hydroxyethyl)Anachidonamide ; N-Arachidonoyl-2-Hydroxyethylamide ; Arachidonoyl Ethanolamide ; Arachidonylethanolamide
Product Categories: Cannabinoid receptor
MF: C22H37NO2
MW: 347.53
Density: 0.94 g/cm3
Flash Point: 269.7 °C
Boiling Point: 522.3 °C at 760 mmHg
Vapour Pressure: 4.33E-13 mmHg at 25°C
Enthalpy of Vaporization: 91.55 kJ/mol
Storage temp.: −20°C
Solubility ethanol: soluble
Following is the molecular structure of Anandamide (CAS NO.94421-68-8) is:

History
Its structure was first described by Czech analytical chemist Lumír Ond?ej Hanuš and American molecular pharmacologist William Anthony Devane in the Laboratory of Raphael Mechoulam, at the Hebrew University in Jerusalem, Israel in 1992. The name is taken from the Sanskrit word ananda, which means "bliss, delight", and amide.
Uses
Anandamide (CAS NO.94421-68-8) has been shown to be involved in working memory.Studies are under way to explore what role it plays in human behavior, such as eating and sleep patterns, and pain relief. It is also important for implantation of the early stage embryo in its blastocyst form into the uterus. Also it is important in the regulation of feeding behavior, and the neural generation of motivation and pleasure. Both anandamide and exogenous cannabinoids like THC enhance food intake in animals and humans, an effect that is commonly known as "the munchies". In addition, anandamide injected directly into the forebrain reward-related brain structure nucleus accumbens enhances the pleasurable responses of rats to a rewarding sucrose taste, and enhances food intake as well. Moreover,it is thought to be an endogenous ligand for vanilloid receptors (which are involved in the transduction of acute and inflammatory pain signals), activating the receptor in a PKC-dependent (protein kinase C-dependent) manner.
Safety Profile
Experimental reproductive effects. When heated to decomposition it emits toxic vapors of NOx.
Hazard Codes:
 F
F
Risk Statements:
R11:Highly flammable.
Safety Statements:
S24/25:Avoid contact with skin and eyes.
S16:Keep away from sources of ignition.
S7:Keep container tightly closed.
RIDADR: UN 1170 3/PG 2
WGK Germany: 3
RTECS: JX3842500
Specification
Anandamide (CAS NO.94421-68-8), known as N-arachidonoylethanolamine or AEA, is an endogenous cannabinoid neurotransmitter found in animal and human organs, especially in the brain. It was isolated. It is degraded by the fatty acid amide hydrolase (FAAH) enzyme which converts anandamide into ethanolamine and arachidonic acid. As such, inhibitors of FAAH lead to elevated anandamide levels and are being pursued for therapeutic use.