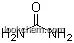Products Categories
| CAS No.: | 7803-63-6 |
|---|---|
| Name: | Ammonium hydrogen sulfate |
| Article Data: | 86 |
| Molecular Structure: | |
|
|
|
| Formula: | H3N . H2 O4 S |
| Molecular Weight: | 115.11 |
| Synonyms: | Ammoniumsulfate ((NH4)HSO4) (6CI); Sulfuric acid, monoammonium salt (8CI,9CI); Acidammonium sulfate; Ammonium acid sulfate; Ammonium bisulfate; Ammonium hydrogensulfate; Ammonium hydrogen sulfate (NH4HSO4); Ammonium monohydrogen sulfate;Monoammonium hydrogen sulfate; Monoammonium sulfate |
| EINECS: | 232-265-5 |
| Density: | 1.79 |
| Melting Point: | 121-145 ºC |
| Boiling Point: | 350 ºC (dec.) |
| Solubility: |
soluble
Stability
Toxicology
|
| Appearance: | white crystalline powder or chunks |
| Hazard Symbols: |

|
| Risk Codes: | R34 |
| Safety: | Moderately toxic by ingestion. A corrosive. See also SULFATES. Dangerous; when heated to decomposition it emits highly toxic fumes of sulfuric acid and SOx, NH3, and NOx. |
| Transport Information: | UN 2506 |
| PSA: | 85.81000 |
| LogP: | 0.46160 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- 90221-55-92-bromo-5-methylbenzaldehyde
- 885590-99-82,3-DIFLUORO-4-IODOBENZALDEHYDE
- 97730-31-9(S)-4'-(2-Methylbutyl)Biphenyl-4-Carbonitrile

| Conditions | Yield |
|---|---|
| With air In solid by standing in air for 24 h; | 99% |

| Conditions | Yield |
|---|---|
| With water heat of evapn., 2 h, 2 M HCl; | A n/a B 67% C n/a |
| With H2O heat of evapn., 2 h, 2 M HCl; | A n/a B 67% C n/a |


| Conditions | Yield |
|---|---|
| In sulfuric acid heating with an excess of H2SO4;; |


| Conditions | Yield |
|---|---|
| With ammonia; water; oxygen; vanadia In further solvent(s) 150-200 °C, in molten equimolar mixture of NH4HSO4 and NaHSO4 dissolving V2O5; transition metal sulfates enhances activity; |

| Conditions | Yield |
|---|---|
| With water; chlorine byproducts: NH4Cl, HCl; with NH3 excess; |

| Conditions | Yield |
|---|---|
| heating at 180°C; |

- 1333-74-0
hydrogen

- 10102-43-9
nitrogen(II) oxide

A
- 227934-46-5
nitrous oxide

B
- 7803-63-6
ammonium bisulfate

C
- 10046-00-1
hydroxylamine sulfate

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal In sulfuric acid Kinetics; 40 °C, P(H2) = 50 kPa, P(NO) = 10 kPa; |


- 7803-63-6
ammonium bisulfate

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water satd. soln. of neutral sulfate is distd. with aq. HCl;; | |
| With sulfuric acid In sulfuric acid cooling a warm solution of (NH4)2SO4 in H2SO4; | |
| Pt boat; 300 to 320 ° C and for a short period at red heat; |

| Conditions | Yield |
|---|---|
| 200 to 250°C; | |
| heating in open vessel; ambient or reduced pressure; | |
| In neat (no solvent) Kinetics; Thermal decomposition.; | |
| 200 to 250°C; | |
| In neat (no solvent) decompn. of (NH4)2SO4 into NH3 and NH4HSO4 at 300-455°C was studied; TG and DSC; |

B
- 7803-63-6
ammonium bisulfate

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: NH3, H2O; 150°C; IR spectroscopy, X-ray diffraction; |

- Total:15 Page 1 of 1 1

What can I do for you?
Get Best Price
Chemistry
IUPAC Name: Azanium hydrogen sulfate
The MF of Azanium hydrogen sulfate (CAS NO.7803-63-6) is H5NO4S.
.gif)
The MW of Azanium hydrogen sulfate (CAS NO.7803-63-6) is 115.11.
Synonyms of Azanium hydrogen sulfate (CAS NO.7803-63-6): Ammonium bisulfate ; Ammonium hydrogensulfate ; Ammonium sulfate monobasic ; Sulfuric acid, monoammonium salt
Product Categories: Inorganics
Apperance: white crystalline powder or chunks
Boiling Point: 330 °C
Melting Point: 146.9 °C
Density; 1.79 g/mL at 25 °C(lit.)
Solubility in H2O: 1 M at 20 °C, clear, colorless
Sensitive: Hygroscopic
Merck: 13,500
Stability: Stable.Hygroscopic
EINECS: 232-265-5
Uses
Azanium hydrogen sulfate (CAS NO.7803-63-6) can be further neutralized with ammonia to form ammonium sulfate, a valuable fertilizer.Also used as analytical reagent and in the pharmaceutical industry.
Consensus Reports
Reported in EPA TSCA Inventory.
Safety Profile
Moderately toxic by ingestion. A corrosive. See also SULFATES. Dangerous; when heated to decomposition it emits highly toxic fumes of sulfuric acid and SOx, NH3, and NOx.Safety information of Azanium hydrogen sulfate (CAS NO.7803-63-6):
Hazard Codes  C
C
Risk Statements
34 Causes burns
Safety Statements
26 In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37/39 Wear suitable protective clothing, gloves and eye/face protection
45 In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
RIDADR UN 2506 8/PG 2
WGK Germany 1
F 3
HazardClass 8
PackingGroup II
Standards and Recommendations
DOT Classification: 8; Label: Corrosive
Specification
Ammonium bisulfate, also known as ammonium hydrogen sulfate, is a white, crystalline solid when it is completely pure, with formula (NH4)HSO4. It is commonly collected as a byproduct of flue gas desulfurization, formed by partial neutralization of sulfuric acid (H2SO4) aerosols by gaseous ammonia, NH3. Indeed, the solid material can collect on and foul solid surfaces in the flues. It is too acidic for most end uses, and is corrosive to the skin.
It react as acids to neutralize bases. These neutralizations generate heat, but less or far less than is generated by neutralization of inorganic acids, inorganic oxoacids, and carboxylic acid. They usually do not react as either oxidizing agents or reducing agents but such behavior is not impossible. Many of these compounds catalyze organic reactions.
When heated to high temperatures, Ammonium hydrogen sulfate may release toxic sulfur oxide and nitrogen oxide fumes. Ammonium hydrogen sulfate is soluble in water. Ammonium hydrogen sulfate is a chemical catalyst, used in hair preparations.